18. A balloon contains 7.2 L of He. The pressure is reduced to
|| 2.00 atm and the
balloon expands to occupy a volume of 25.1 L. What was
the initial pressure
exerted on the balloon?
Solved
Show answers
More tips
- C Computers and Internet Best Antivirus: How to Choose Protection for Your Computer?...
- L Legal consultation What Documents Are Required for a Russian Passport?...
- S Sport How to Choose Tennis Rackets?...
- H Health and Medicine AKDS Vaccination: Ensure Your Child s Safety...
- H Health and Medicine Naskol ko Opasen Ukus Kleshcha i Kak Ego Raspoznat...
- C Computers and Internet How to Delete Your Account on Odnoklassniki...
- H Health and Medicine What to Do When Your Jaw Locks Up?...
- G Goods and services What Are the Most Popular Services?...
- P Philosophy How did the concept of module arise in computer science?...
Answers on questions: Chemistry
- C Chemistry Which substance cannot be broken down by chemical changes? Magnesium Hydroxide Carbon Dioxide b O Ammonia Iron...
- C Chemistry How many moles are equal to 7.12x10e24 atoms of aluminum?...
- C Chemistry An example of a chemical change is a) dissolving sugar in water b) sanding wood c) gasoline burning d) melting ice and explain plz so i can rlly learn it !...
- C Chemistry Which of the following elements is the most reactive? beryllium calcium magnesium barium...
- C Chemistry Please help me with this question please help...
- C Chemistry Please help me with questions 2 please help...
- M Mathematics \log_{4} x + \log_{4} (x+5) = 6...
- M Mathematics 4.2meters= how many centimeter...
- M Mathematics 31+34=90-n 45+1=70-k 6×9=41+m...
- M Mathematics 5y-3x+2=0 to y=mx+b form...

Ответ:
The initial pressure exerted on the balloon is 7.0 atm.
Explanation:
To calculate the new volume, we use the equation given by Boyle's law. This law states that pressure is inversely proportional to the volume of the gas at constant temperature.
The equation given by this law is:
where,
We are given:
Initial pressure of helium gas in balloon =
Initial volume of helium gas in balloon =
Final pressure of helium gas in balloon =
Final volume of helium gas in balloon =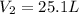
Putting values in above equation, we get:
Hence, the initial pressure exerted on the balloon is 7.0 atm.
Ответ:
ur cute
Explanation: