josephbrowne9p18dit
17.09.2019 •
Chemistry
2.3 zinc has five naturally occurring isotopes: 48.63% of 64 zn with an atomic weight of 63.929 amu; 27.90% of 66zn with an atomic weight of 65.926 amu; 4.10% of 67zn with an atomic weight of 66.927 amu; 18.75% of 68zn with an atomic weight of 67.925 amu; and 0.62% of 70zn with an atomic weight of 69.925 amu. calculate the average atomic weight of zn.
Solved
Show answers
More tips
- C Computers and Internet How to Delete a Page on VKontakte: The Ultimate Guide from Experts...
- S Style and Beauty 5 Tips for Choosing the Best Ugg Boots...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- H Health and Medicine Effective Ways to Treat Colic in Infants...
- C Computers and Internet IMHO: What is it, why is it important, and how to use it?...
- F Food and Cooking How to Calculate the Gender of Your Child with Blood?...
- S Style and Beauty How to Get Rid of Under Eye Bruises?...
- C Cities and Countries What time does the Metro open in Moscow?...
- W Work and Career Where can you learn to be a flight attendant?...
- H Health and Medicine What are Electronic Cigarettes? The Ultimate Guide...
Answers on questions: Chemistry
- S Spanish John Lewis dedicó su vida a proteger los derechos de los negros. ¿Qué opinas de esta cita de John Lewis? Nada puede detener el poder de un pueblo comprometido y decidido a lograr...
- H Health Describe at least 2 impacts of noncommunicable diseases on people and society...
- H History In what ways did the Stalin purges violate the concept of due process of law ?...
- E English Ming s mother fell asleep on the couch right after dinner. Ming didn t blame her - she worked long hours. She had an important job at a big company that Ming didn t really understand....

Ответ:
The average atomic mass of element Zinc is 65.40 amu.
Explanation:
Average atomic mass of an element is defined as the sum of masses of each isotope each multiplied by their natural fractional abundance.
Formula used to calculate average atomic mass follows:
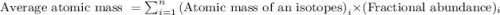 .....(1)
.....(1)
ForMass of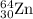 isotope = 63.929 amu
isotope = 63.929 amu
Percentage abundance of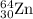 isotope = 48.63 %
isotope = 48.63 %
Fractional abundance of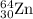 isotope = 0.4863
isotope = 0.4863
ForMass of isotope = 65.926 amu
isotope = 65.926 amu
Percentage abundance of isotope = 27.90 %
isotope = 27.90 %
Fractional abundance of isotope = 0.2790
isotope = 0.2790
ForMass of isotope = 66.927 amu
isotope = 66.927 amu
Percentage abundance of isotope = 4.10 %
isotope = 4.10 %
Fractional abundance of isotope = 0.0410
isotope = 0.0410
ForMass of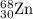 isotope = 67.925 amu
isotope = 67.925 amu
Percentage abundance of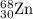 isotope = 18.75 %
isotope = 18.75 %
Fractional abundance of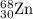 isotope = 0.1875
isotope = 0.1875
ForMass of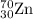 isotope = 69.925 amu
isotope = 69.925 amu
Percentage abundance of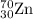 isotope = 0.62 %
isotope = 0.62 %
Fractional abundance of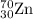 isotope = 0.0062
isotope = 0.0062
Putting values in equation 1, we get:
Hence, the average atomic mass of element Zinc is 65.40 amu.
Ответ: