kamila20394
31.07.2019 •
Chemistry
2. consider the reaction 2no(g) + o2(g) → 2no2(g) suppose that at a particular moment during the reaction nitric oxide (no) is reacting at the rate of 0.066 m/s. a. at what rate is no2 being formed? (3 points) b. at what rate is molecular oxygen reacting? (3 points)
Solved
Show answers
More tips
- C Computers and Internet Are there special gaming mice?...
- L Leisure and Entertainment When will Maslenitsa start?...
- F Food and Cooking Discovering the Mysterious Fruit of Feijoa...
- B Business and Finance How to Open an Online Store? A Detailed Guide for Beginners...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
Answers on questions: Chemistry
- C Chemistry How many significant figures are in the number 0.00230? o 6...
- C Chemistry What is accuracy? the same as precision it refers to how closely individual measurements agree with each other. it referes to how closely a measured value agrees with the correct...
- C Chemistry What does 1.23 + 45.6 equal? 46.9 o 47 o 46.8 o 46.83...
- C Chemistry What is the proper scientific notation for 1234? o 1.234x103 o 0.1234x104 o 1.234x102 o 12.34x102...
- C Chemistry How many significant figures are in the number 230? o2 01 o 23...
- C Chemistry How many significant figures are in the number 230.0? 04 nw...
- C Chemistry Commercial nitric acid comes in a concentration of 16.0 mol/l. the density of this solution is 1.42 g/ml calculate the percent (w/w) of nitric acid, hno3, in this solution. how many...
- C Chemistry How many nanoseconds are in 1 megasecond? o 1.0 x 1015 seconds o 1.0 x 1021 seconds o 1.0 x 10-15 seconds 1.0 x 1012 seconds...
- C Chemistry Am radio signals broadcast electromagnetic radiation at frequencies between 640 khz and 1.20 mhz. (a) what is the shortest wavelength of an am radio signal? in m (b) what is the...
- C Chemistry Which equation can be used to convert from celsius to kelvin? o celsius + 459.67 o (celsius)x1.8+32 o (celsius-32)x5/9 celsius+273.15...

Ответ:
Answer :
(a) The rate of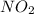 formed is, 0.066 M/s
formed is, 0.066 M/s
(b) The rate of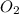 formed is, 0.033 M/s
formed is, 0.033 M/s
Explanation : Given,
The balanced chemical reaction is,
The rate of disappearance of =
= ![-\frac{1}{2}\frac{d[NO]}{dt}](/tpl/images/0155/9230/9766a.png)
The rate of disappearance of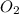 =
= ![-\frac{d[O_2]}{dt}](/tpl/images/0155/9230/eb70c.png)
The rate of formation of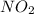 =
= ![\frac{1}{2}\frac{d[NO_2]}{dt}](/tpl/images/0155/9230/de302.png)
As we know that,
(a) Now we have to determine the rate of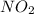 formed.
formed.
The rate of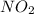 formed is, 0.066 M/s
formed is, 0.066 M/s
(b) Now we have to determine the rate of molecular oxygen reacting.
The rate of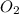 formed is, 0.033 M/s
formed is, 0.033 M/s
Ответ: