robertss403
20.03.2020 •
Chemistry
2 HI(g) ⇄ H2(g) + I2(g) Kc = 0.0156 at 400ºC 0.550 moles of HI are placed in a 2.00 L container and the system is allowed to reach equilibrium. Calculate the concentration of H2 at equilibrium. 0.0275 M (Your correct answer) 0.138 M 0.275 M 0.550 M 0.220 M
Solved
Show answers
More tips
- A Animals and plants How to Grow Lime from a Seed: Simple Tips and Interesting Facts...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
Answers on questions: Chemistry
- C Chemistry Which energy conversion occurs in an operating voltaic cell? A) electrical energy to nuclear energy B) chemical energy to electrical energy C) chemical energy to nuclear...
- C Chemistry Which property describes the way substances react with other substances to form new substances?...
- C Chemistry To make its second-highest energy level be its valence energy level (being filled with an octet of electrons) and possess a more stable electronic arrangement, a strontium...
- C Chemistry What is the mass number of an atom that contains 12 protons, 14 neutrons, and 12 electrons? * 10 points 2 26 24 168...
- C Chemistry Pls helppp How many energy must be absorbed by 150.0 grams of water to raise it s temperature by 50.00°C?...
- H Health The concept that a healthcare provider should do good for the patient....
- S Social Studies The most common reason that people leave their jobs is...
- S Social Studies What are the key compromises and provisions in the us constitution...
- M Mathematics Given a rectangular sheet of paper 8.5 inches by 11 inches, form a box by cutting congruent squares from each corner, folding up the sides, and taping them to form a box...
- S Social Studies The process in which members of one cultural group adopt the beliefs and behaviors of another group....

Ответ:
The concentration of hydrogen gas at equilibrium is 0.0275 M
Explanation:
Molarity is calculated by using the equation:
Moles of HI = 0.550 moles
Volume of container = 2.00 L
For the given chemical equation:
Initial: 0.275
At eqllm: 0.275-2x x x
The expression of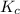 for above equation follows:
for above equation follows:
We are given:
Putting values in above expression, we get:
Neglecting the negative value of 'x' because concentration cannot be negative
So, equilibrium concentration of hydrogen gas = x = 0.0275 M
Hence, the concentration of hydrogen gas at equilibrium is 0.0275 M
Ответ:
6.68%
Explanation:
FV= 1000
N = 25*2=50
PMT= 8.75%*1000/2= 43.75
PV = -1250
I/Y = RATE(25*2, 8.75%*1000/2, -1200, 1000)
I/Y = 0.033397
I/Y = 3.34%
YTM = 3.34% * 2
YTM = 6.68%