sierrabuckner397
13.05.2021 •
Chemistry
26 grams of 2,2 dimethyl hexane( C8H16) under goes combustion. How much space would the CO2 gas occupy at 26 Celsius and 101.75 kPa?
Solved
Show answers
More tips
- C Computers and Internet What is the Meaning of lol and How Did it Become Popular?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- H Health and Medicine How to Help Men Gain Weight?...
- F Family and Home How to Build a Strong Relationship with Your Child: Tips for Effective Communication...
- L Legal consultation Juvenile Justice: Who Needs It?...
- F Food and Cooking What age is appropriate for giving your child cocoa?...
- A Animals and plants Why do cats go crazy over catnip?...
- H Health and Medicine How to Cure Adenoids?...
- S Science and Technology Why is there no gravity on other planets?...
- F Food and Cooking How to Properly Wash a Down Jacket? Tips from Experts...
Answers on questions: Chemistry
- S Spanish PLEASE HELLPPPP PLEASEEEEEE...
- H History If the robber is not caught, the man who has been robbed shall formally declare whatever he has lost before a god, and the city and the mayor in whose territory or district the robbery...
- H History Republicans argued that high tariffs would...
- E English PLEASE HELP ME Caro is revising her narrative and focusing on her opening paragraph.Captain Bernard braced himself against the ship s sturdy mast. There was no turning back. The storm...
- E English Summarize The Hobbit thus far. What has happened? Who are the characters? Where did they go? What are they doing? It must be between 5-7 sentences. page 112 to 126 please help...

Ответ:
The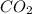 gas occupies a space of 5.57 L at 26 Celsius and 101.75 kPa.
gas occupies a space of 5.57 L at 26 Celsius and 101.75 kPa.
Explanation:
Given: Mass = 26 g
Pressure = 101.75 kPa
Convert kPa into atm as follows.
Temperature =
Now, moles of a substance is defined as mass of substance divided by its molar mass.
As molar mass of 2,2 dimethyl hexane is 114.23 g/mol. So, its number of moles are calculated as follows.
Formula used to calculate the volume is as follows.
PV = nRT
where,
P = pressure
V = volume
n = no. of moles
R = gas constant = 0.0821 L atm/mol K
T = temperature
Substitute the values into above formula as follows.
Thus, we can conclude that the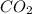 gas occupies a space of 5.57 L at 26 Celsius and 101.75 kPa.
gas occupies a space of 5.57 L at 26 Celsius and 101.75 kPa.
Ответ: