3. Commercial phosphoric acid, H3PO4, is often purchased as a 85.5 weight percent solution. Find the mg/L of H2SO4 and the molarity and normality of the solution. The concentrated sulfuric acid solution has a density of 1.71 g/ml at the temperature of the system.
Solved
Show answers
More tips
- S Sport Where can you play football in Moscow?...
- F Food and Cooking 10 Reasons Why You Should Avoid Giving Re-Gifts: An Informative Guide...
- S Sport How to wrap boxing hand wraps? Everything you need to know!...
- A Animals and plants 5 Tips for Taking Care of Yews to Keep Them Green and Beautiful...
- H Health and Medicine How to Calm Your Nerves? Expert Tips That Actually Work...
- O Other What is a Disk Emulsifier and How Does it Work?...
- S Sport How to Pump Your Chest Muscle? Secrets of Training...
- C Computers and Internet How to Get Rid of 3pic Infector: Everything You Need to Know...
- S Style and Beauty How to Grow Hair Faster: Real Methods and Advice...
- C Computers and Internet How to Top Up Your Skype Account Without Losing Money?...

Ответ:
The answers to the question are
(a) The the mg/L of H₃PO₄is 1462.05 g
(b) The molarity of H₃PO₄ in the solution is 14.92 mol/L
(c) The normality of H₃PO₄ in the solution 44.85 eq/L
Explanation:
(a) The commercially available H₃PO₄ has a concentration of 85.5 percent by weight, therefore
The mass of H₃PO₄ is found by
Density of water = 1.71 g/ml
mass of one liter of acid solution = 1.71 ×1000 = 1710 g
Therefore 85.5 % by weight of H₃PO₄ =1710×85.5/100 = 1462.05 g
Therefore we have 1462.05 g/L of H₃PO₄
Molar mass of H₃PO₄ = 97.994 g/mol
(b)Therefore the number of moles in 1462.05 g = 14.92 moles and the molarity = 14.92 mol/L
(c) The Normality =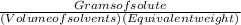 = 1465.05÷(1×97.994/3) = 44.85 eq/L
= 1465.05÷(1×97.994/3) = 44.85 eq/L
Ответ:
First, he should measure how long it takes for the liquid to become clear if X and Y are mixed together. Then, he should measure how long it takes if he also adds substance A to X and Y. He will find out if substance A is a catalys
Explanation:
did the test rn