3) What is the molarity of a solution that has 27.6 g of propanol (C:H:O) dissolved in 325 ml. ot solution? The molar mass of ethanol is 46.08 g/mol
Solved
Show answers
More tips
- C Computers and Internet IMHO: What is it, why is it important, and how to use it?...
- H Health and Medicine How to Calculate Your Ideal Weight?...
- S Style and Beauty Discover the Art of Nail Design: How Do You Paint Your Nails?...
- P Philosophy How to Develop Extrasensory Abilities?...
- O Other Everything You Need to Know About Kudyabliks...
- C Computers and Internet The Twitter Phenomenon: What it is and How to Use it...
- C Computers and Internet How to Choose a Laptop: Expert Guide and Tips...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
Answers on questions: Chemistry
- C Chemistry Why were the proton and electron discovered before the neutron...
- C Chemistry 3. if you have had a clogged sink drain at your home, you have undoubtedly tried using a commercial drain cleaner to dissolve the clog. what evidence is there that...
- C Chemistry How many atoms of fluorine are in ZrF4...
- C Chemistry 10.stephanie observes the color of a liquid.then she uses a thermometer to measure the temperature of the liquid. finally, she uses a graduated cylinder to find the...
- M Mathematics 1. match each function with the correct table, graph, or description. 2.drag equations to graph. 3. complete the table for f(x) = x2 - 2 third pic goes with question...
- M Mathematics Find the percent increase or decrease to the nearest percent. from 14 to 40 books. type whether the percent change is an increase or decrease...
- M Mathematics What is the first step to 2x + 3y = 12?...
- M Mathematics Find the measure of angle x in the figure below...
- M Mathematics How do you solve x divided by 7 + x equals 8?...
- M Mathematics What are the constants for the problem 7 - 5x + 1...

Ответ:
The empirical and molecular formula for the given organic compound is CH and and it is not an alkane.
and it is not an alkane.
Explanation:
The chemical equation for the combustion of hydrocarbon having carbon and hydrogen follows:
where, 'x' and 'y' are the subscripts of Carbon, hydrogen and oxygen respectively.
We are given:
Mass of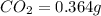
Mass of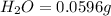
We know that:
Molar mass of carbon dioxide = 44 g/mol
Molar mass of water = 18 g/mol
For calculating the mass of carbon:
In 44 g of carbon dioxide, 12 g of carbon is contained.
So, in 0.364 g of carbon dioxide, of carbon will be contained.
of carbon will be contained.
For calculating the mass of hydrogen:
In 18 g of water, 2 g of hydrogen is contained.
So, in 0.0596 g of water,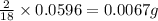 of hydrogen will be contained.
of hydrogen will be contained.
To formulate the empirical formula, we need to follow some steps:
Step 1: Converting the given masses into moles.Moles of Carbon =
Moles of Hydrogen =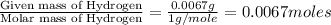 ]
]
Step 2: Calculating the mole ratio of the given elements.For the mole ratio, we divide each value of the moles by the smallest number of moles calculated which is 0.0067 moles.
For Carbon =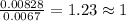
For Hydrogen =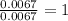
Step 3: Taking the mole ratio as their subscripts.The ratio of C : H = 1 : 1
The empirical formula for the given compound is CH
For determining the molecular formula, we need to determine the valency which is multiplied by each element to get the molecular formula.
The equation used to calculate the valency is:
We are given:
Mass of molecular formula = 128.2 g/mol
Mass of empirical formula = 13 g/mol
Putting values in above equation, we get:
Multiplying this valency by the subscript of every element of empirical formula, we get:
The general formula of an alkane is , where n = any natural number
, where n = any natural number
Here, n = 10 and it does not satisfy being an alkane
Hence, the empirical and molecular formula for the given organic compound is CH and and it is not an alkane.
and it is not an alkane.