36) which pair represents two forms of an element in the same phase at stp but with different structures and different properties? a) i2(s) and i2(g) b) o2(g) and o3(g) c) h2(g) and hg(g) d) h2(s) and h2o()
Solved
Show answers
More tips
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- H Health and Medicine Headache: A Comprehensive Guide to Treatment...
- C Computers and Internet How to Properly Order Clothing from International Online Stores...
- A Auto and Moto Mastering One-Movement Parking: All You Need to Know...
- D Dating, Love, Relationships How Long Can Love Last?...
- S Society and Politics Is It Fact or Fiction? Let s Talk About Anton Chekhov s Pseudonym...
- F Food and Cooking How to Find Your Zip Code?...
- S Style and Beauty How to choose the best mascara for your eyelashes...
- S Style and Beauty How to Apply Foundation Correctly?...
- S Sport How to Choose Tennis Rackets?...
Answers on questions: Chemistry
- M Mathematics Select the values that make the inequality h −7 true....
- M Mathematics I really need help solving this math problem. -6-5x^2+7x^2-7y^3+y^3+4+2x^2...
- E English And this is from the story of gluskabe and old man winter. why does gluskabe need the balls of a.to fool the crows into grabbing a ball instead of his head b. to...

Ответ:
The correct answer is option B.
Explanation:
Oxygen element exists in two forms:
1) Oxygen gas :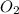
In this two oxygen atoms are bonded to each other with the help of double bond.
2) Ozone =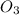
In ozone molecule, two oxygen are linked with double bond and the third oxygen atom is linked to one of the oxygen atom by accepting its lone pair of electrons. Due to this double bond resonates between the ozone molecule by which partial double bond character establishes between oxygen-oxygen bond.
Ответ:
Explanation:
# of protons: 6
# of neutrons: 6
# of electrons: 2, 4
mass: 12.01 grams per mole of carbon atoms.
For the screenshot search up carbon atom and use a picture