djennings8055
24.01.2020 •
Chemistry
371. mg of an unknown protein are dissolved in enough solvent to make 5.00 ml of solution. the osmotic pressure of this solution is measured to be at 0.118 atm at 25 c
calculate the molar mass of the protein.
Solved
Show answers
More tips
- F Food and Cooking 10 Ideas for a Wedding Anniversary Gift...
- H Health and Medicine Hangover: How to Get Rid of It Quickly?...
- S Style and Beauty How to Choose the Right Fur Coat and Avoid Regrets?...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- P Physics What is the frequency of a sound wave that is traveling through water at 1400 m/s and has a wavelength of 7.0 m? * f= 07.0 Hz O700 Hz О 200 Hz ОО.01 Hz...
- E English Which quotation provides the best evidence for the central idea of this excerpt? In this house Trujillo is Chief. The pale face of a young military man wearing a plumed...

Ответ:
The molar mass of the protein is 15394.2 g/mol
Explanation:
To calculate the concentration of solute, we use the equation for osmotic pressure, which is:
where,
i = Van't hoff factor = 1 (for non-electrolytes)
M = molarity of solute = ?
R = Gas constant =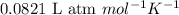
T = temperature of the solution =![25^oC=[273+25]=298K](/tpl/images/0468/1226/6a9f9.png)
Putting values in above equation, we get:
To calculate the molecular mass of solute, we use the equation used to calculate the molarity of solution:
We are given:
Molarity of solution =
Given mass of protein = 371. mg = 0.371 g (Conversion factor: 1 g = 1000 mg)
Volume of solution = 5.00 mL
Putting values in above equation, we get:
Hence, the molar mass of the protein is 15394.2 g/mol
Ответ:
(-1,0)?
Step-by-step explanation: