clairajogriggsk
12.03.2021 •
Chemistry
4. A large quantity of a reactant that precipitates solid gold from gold in solution was added
to a large sample of ocean water, but only a trace amount of solid gold resulted after
filtering the mixture. What do you think could be the problem? Describe one of the reactants
as a limiting reagent in your answer.
Solved
Show answers
More tips
- S Society and Politics Как правильно поддерживать температуру в квартире для здоровья...
- L Leisure and Entertainment How to Make a Crab Trap in Just a Few Minutes...
- H Health and Medicine How Much Does Abortion Cost? Expert Answers and Insights...
- S Sport How to Build Arm Muscles? Effective Exercises and Tips...
- H Health and Medicine When can it be said that a person has a normal pulse?...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
Answers on questions: Chemistry
- C Chemistry Acids produce ions when dissolved in water. acids react with many to produce gas....
- C Chemistry Calculate the approximate mass of naoh required to prepare 500 ml of a 0.15 m naoh solution...
- C Chemistry What is the equilibrium constant expression for the given system? o keat h: 102] [h20] [h2012 o kegf th; p[02] [hz]? [02] reg- theo] [h21? [02]...
- C Chemistry How does describing how ideas in science texts are connected help you understand how things work and why things change?...
- C Chemistry Charcoal can be soaked in lighter fluid when grilling food. Lighter fluid is extremely flammable. This is an example of which type of property? Physical property Chemical...
- C Chemistry How many kilojoules of heat are required to vaporize 50.0 g of ethanol (MM = 46.07 g). Its molar heat of vaporization is 43.5 kJ/mol....
- C Chemistry Segun la cantidad de atomos de un ion ¿como se llama?...
- C Chemistry Please help, what does this mean?...
- C Chemistry What does the difference in potential energy between the reactants and products represent? A. It represents the energy required to make the reaction happen. B. It represents...
- C Chemistry When aluminum reacts with concentrated hydrochloric acid, hydrogen gas is produced. 2Al(s)+6HCl(aq)⟶2AlCl3(aq)+3H2(g) What mass of Al(s) is required to produce 560.0...

Ответ:
Ответ:
a. 249.5 g/mol
b. 1.996 g
c. Measure 1.996 g of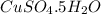 and dissolve in water until the volume is 400 ml
and dissolve in water until the volume is 400 ml
Explanation:
Molar mass is the sum of atomic masses of all the elements present.
Thus molar mass of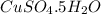 = 1(63.5)+1(32)+4(16)+5(18) = 249.5 g/mol
= 1(63.5)+1(32)+4(16)+5(18) = 249.5 g/mol
Molarity of a solution is defined as the number of moles of solute dissolved per liter of the solution.
To calculate the number of moles for given molarity, we use the equation:
c. We need to measure 1.996 g of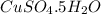 and dissolve it in water to make the volume 400 ml.
and dissolve it in water to make the volume 400 ml.