hannaharsena
20.04.2020 •
Chemistry
5.943x10^24 molecules of H3PO4 will need how many grams of Mg(OH)2 in the reaction below? 3 Mg(OH)2 + 2 H3PO4 > 1 Mg3(PO4)2 + 6 H2O
Solved
Show answers
More tips
- G Goods and services How to Choose a Video Camera: Tips from Professionals...
- F Family and Home How to Remove Tar Stains: Tips and Recommendations from Experts...
- H Health and Medicine Novomin: What is it and how to use it?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...

Ответ:
5.943 × 10²⁴ molecules of H₃PO₄ will react with 789.29 grams of Mg(OH)₂
Explanation:
Here we have
5.943 × 10²⁴ molecules of H₃PO₄ in a reaction with Mg(OH)₂
as follows
3Mg(OH)₂ + 2H₃PO₄ → Mg₃(PO₄)₂ + 6H₂O
Therefore 3 moles of Mg(OH)₂ react with 2 moles of H₃PO₄ to form 1 mole of Mg₃(PO₄)₂ and 6 moles of H₂O
5.943 × 10²⁴ molecules of H₃PO₄ which is equivalent to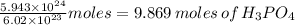
Will react with 3/2×9.869 moles or 14.8 moles of Mg(OH)₂
One mole of Mg(OH)₂ weighs 58.3197 g/mol
Therefore, 5.943 × 10²⁴ molecules of H₃PO₄ will react with 14.8×58.3197 g or 789.29 grams of Mg(OH)₂.
Ответ:
Because Polar regions get less of the sun's direct rays, they are the coldest climates on Earth.
Explanation: