morgandodd2001
15.05.2021 •
Chemistry
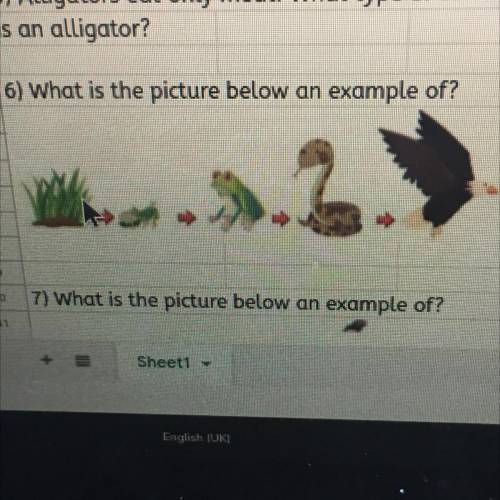
6) What is the picture below an example of?
Solved
Show answers
More tips
- S Style and Beauty What Peeling is Suitable for Sensitive Skin?...
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- C Chemistry What is the ph of a 0.051 m solution of hcl?...
- C Chemistry Explain collision theory and discuss how it can be used to explain why powdered sugar will dissolve in water faster than a cube of sugar....
- C Chemistry What kind of crystalline solid is nickel (Ni)? O A. Molecular solid O O O B. Ionic solid C. Metallic solid D. Network solid...
- C Chemistry At equilibrium the partial pressures of N2O4 and NO2 are 0.35 atm and 4.3 atm. What is the Kp...
- C Chemistry A car turns into star street does this mean the cars velocity changed...
- C Chemistry Using only the periodic table, determine the element that displays each given trend. be sure to provide your reasoning. choose the element that is more reactive: oxygen...
- C Chemistry Some please please help me with this question...
- C Chemistry What is the mass of 100 mL of corn oil?...
- M Mathematics 6th grade math help me, please :)...
- M Mathematics The base of a regular pyramid is a hexagon....

Ответ:
The picture below is an example of a food chain.
Explanation:
The bug (can't tell which one by the picture) eats the grass, the frog eats the bug, the snake eats the frog, and the eagle eats the snake.
Ответ:
Ответ:
The heat produced by burning of 1.9 mole of methane gas will be 1691.38 kJ.
Explanation: Standard molar enthalpy of combustion is defined as the change when 1 mole of the hydrocarbon gets completely burnt in the presence of oxygen gas at standard temperature and pressure.
Equation for the combustion of methane gas is given by:
We are given standard molar enthalpy of combustion for methane gas = 890.2 kJ/mol
This is the heat required for 1 mole of a gas.
So, heat required for 1.9 moles of a gas = 890.2kJ/mol × 1.9mol = 1691.38 kJ