almostnevercbbc
04.07.2019 •
Chemistry
7.00g of ech reactant is available, determine the theoretical yield of 2hg(l) + o2(g) - 2 hgo
Solved
Show answers
More tips
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
- P Photography and Videography How to Choose the Perfect Photo Paper for Your Images?...
Answers on questions: Chemistry
- C Chemistry How many valence electrons does carbonic acid contain?...
- C Chemistry How do the prefixes in the metric system relate to the basic units?...
- C Chemistry 8) C(diamond) → C(graphite) delta G° = -2.9 kJ/molrxn Which of the following best explains why the reaction represented above is not observed to occur at room temperature? (A)...
- C Chemistry Can someone pls help me with this :)...
- C Chemistry How many grams are needed to make 675 mL of 0.980 M solution of Calcium chloride, CaCl2...
- C Chemistry What is the [H30 ] in a solution of pH 0.60?...
- C Chemistry Can somebody plz help answer all of these questions right it’s very important. If u can that will be amazing thanks! WILL MARK BRAINLIEST WHOEVER ANSWERS FIRST :DDD...
- P Physics Which chemical equation is unbalanced? A ) K + H2O -- K2O + H2 B) MgO + 2HCI -- MgCI2 + H2O C) CaO2 + HCI -- CaCI2 + H2O D) CO2 + H2O -- H2CO2 (B Is the correct answer)...
- M Mathematics Select the correct answer Which matrix is the product of a 3 x 3 identity matrix and the scalar 3? O A. 3 3 3 3 3 3 3 3 3 B. OC. 3 0 0 0 3 0 0 0 3 OD 3 1 1 1...
- M Mathematics Draw the image of ABC under a dilation whose center is P and scale factor is 4...

Ответ:
7.88 g
Explanation:
We have the masses of two reactants, so this is a limiting reactant problem.
We know that we will need a balanced equation with masses, moles, and molar masses of the compounds involved.
Step 1. Gather all the information in one place with molar masses above the formulas and masses below them.
2Hg + O₂ ⟶ 2HgO
Mass/g: 7.00 7.00
Step 2. Calculate the moles of each reactant
Step 3. Identify the limiting reactant
Calculate the moles of HgO we can obtain from each reactant.
From Hg:
From O₂: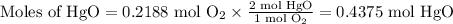
The limiting reactant is Hg because it gives the smaller amount of HgO.
Step 4. Calculate the mass of HgO that you can obtain from Hg.
Ответ:
4x - 2 = 6
Then x = 2