safiyabrowne7286
17.03.2020 •
Chemistry
A 1.00 L flask is filled with 1.15 g of argon at 25 ∘C. A sample of ethane vapor is added to the same flask until the total pressure is 1.350 atm . Part A What is the partial pressure of argon, PAr, in the flask?
Solved
Show answers
More tips
- S Science and Technology Exploring Our Galaxy: How Many Planets are in the Milky Way?...
- W Work and Career Can Skill Alone Make You a Professional?...
- C Computers and Internet How to Top Up Your Skype Account Without Losing Money?...
- P Philosophy Unidentified Flying Object - What is the Nature of this Phenomenon?...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
Answers on questions: Chemistry
- C Chemistry Identify which statements about denaturation are true and which are false....
- C Chemistry A gas expands from an initial volume of 30.0 L to a final volume of 65.0 L at a constant pressure of 110 kPa. How much work is done by the gas during this expansion...
- C Chemistry Jill needs to test her pool to make sure it has enough chlorine in it. She put water in her test tube and then added 5 drops of the test solution. The water turned light pink. This...
- C Chemistry The following reaction is exothermic. 2 N2O(g) 2 N2(g) O2(g) This means the reaction: 56) A) will be spontaneous only at low temperatures B) is not spontaneous at any temperature C)...
- C Chemistry A hemorrhoidal ointment contains, on a weight-in-weight basis, 46.6% mineral oil, 1% pramoxine HCl, and 12.5% zinc oxide in an ointment base. Calculate the grams of each ingredient,...
- C Chemistry As a saturated solution cools, some solute comes out of the solution and falls to the bottom of the container. This is called . A. titration B. evaporation C. dissolution D. precipitation...
- C Chemistry What is the meaning of electrolysis?...
- C Chemistry List the number of protons, neutrons, and electrons in iodine-127. (atomic number is 53)...
- C Chemistry All of the following are examples of carbohydrates except fiber. proteins. starches. sugars....
- C Chemistry The molality of a solution that is made by dissolving a certain mass of ethanol in 85.4 g of water is 0.950 m. how many moles of ethanol were dissolved? use mc028-1.jpg. 0.0811 mol...

Ответ:
The partial pressure of argon in the flask = 71.326 K pa
Explanation:
Volume off the flask = 0.001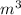
Mass of the gas = 1.15 gm = 0.00115 kg
Temperature = 25 ° c = 298 K
Gas constant for Argon R = 208.13
From ideal gas equation P V = m RT
⇒ P =
Put all the values in above formula we get
⇒ P =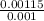 × 208.13 × 298
× 208.13 × 298
⇒ P = 71.326 K pa
Therefore, the partial pressure of argon in the flask = 71.326 K pa
Ответ:
vowels = 0
consonants = 0
string = input("Enter string: ")
for x in string:
if x.isalpha():
if x in "AEIOU":
vowels += 1
else:
consonants += 1
print("Vowels =",vowels)
print("Consonants =",consonants)
Explanation: