A 100.0g sample of tin is heated to 100.0 oC (Celsius) and is placed in a coffee cup calorimeter containing 150. g of water at 25.0 oC. After the metal cools, the final temperature of the metal and the water is 27.4 oC. Calculate the specific heat capacity of tin from these experimental data, assuming that no heat escapes to the surroundings or is transferred to the calorimeter. Specific heat of water
Solved
Show answers
More tips
- G Goods and services How to Choose a Video Camera: Tips from Professionals...
- F Family and Home How to Remove Tar Stains: Tips and Recommendations from Experts...
- H Health and Medicine Novomin: What is it and how to use it?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
Answers on questions: Chemistry
- M Mathematics Question 4 (3 points) If y=4x, is an equation for a different walker, what is their walking rate? per Word Bank: feet meters miles meters second minute second hour Blank 1: Blank...
- H History Why was Kennewick Man an important archaeological find?...
- M Mathematics Ahiker leaves camp and hikes 9 miles north and than hikes 7 miles east. what is the resulting displacement of the hiker?...

Ответ:
Explanation:
It is known that specific heat of water is 4.184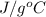 and atomic mass of tin is 118.7 g/mol. For the given situation,
and atomic mass of tin is 118.7 g/mol. For the given situation,
Let us assume that,
Therefore, heat energy expression for heat lost and gained is as follows.
= 0.207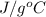
For, 118.7 g the specific heat of tin will be calculated as follows.
= 24.5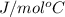
Thus, we can conclude that specific heat of tin is 24.5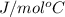 .
.
Ответ:
A. Angles 1 and 5 are congruent corresponding angles
Step-by-step explanation: