kidre96521
11.03.2020 •
Chemistry
A 101 g piece of aluminum (aluminum = 0.900 J/g oC) is heated to 100.1 oC and added to 48.9 g of water at an initial temperature of 16.5oC. what will the final temperature of the mixture be
Solved
Show answers
More tips
- S Science and Technology The Metric System in Our Daily Life: Understanding Its Importance...
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- C Computers and Internet How to Learn to Type Fast?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- S Style and Beauty How to braid friendship bracelets?...
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking Which Calamari Salad is the Most Delicious?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- H Health and Medicine What is Autism? Understanding the Basics of This Neurodevelopmental Disorder...
Answers on questions: Chemistry
- C Chemistry If an object has a density of 8.65 g/cm3, what is its density in units of kg/m3?...
- C Chemistry Does polonium lend or borrow electrons?...
- C Chemistry Cho 3,04 gam hỗn hợp A gồm bột Fe3O4 và FeO vào dung dịch H,SO4 đặc, nóng dư. Sau khi phản ứng xảy ra hoàn toàn thu được 0,224L khí SO2 (đktc). Tính khối lượng mỗi chất...
- C Chemistry Cho m gam hỗn hợp Cu và Fe tác dụng vừa đủ với 1,456lít khí Cl2 (đktc), thu được 7,575g hỗn hợp muối a) tính m gamb) tính thành phần phần trăm theo khối lượng mỗi kim...
- C Chemistry Calculate the heat given off when 177 g of copper cools from 155.0°C to 23.0°C. The specific heat of copper is 0.385 J/gºC....
- C Chemistry What is the energy in joules of a single photon of light with a wavelength of 1 m...
- C Chemistry 4) If the actual measurement is 1.85 mL, write 4 measurements that demonstrate both accuracy and precision Data in mL Trial 1 2 3 4 DS 6) Identify the number of significant...
- M Mathematics Simplify the equation:...
- M Mathematics Will mark brainliest!Only question 2 and 5...
- H History Why is the right of freedom of speech and press protected in this country?...

Ответ:
The final temperature of the mixture is = 42.14 °C
Explanation:
Mass of the aluminium = 101 gm
= 101 gm
Specific heat for aluminium = 0.9
= 0.9 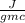
Temperature of aluminium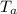 = 100.1 °C
= 100.1 °C
Mass of water = 48.9 gm
= 48.9 gm
Specific heat for water = 4.2
= 4.2 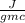
Initial temperature of water = 16.5 °C
= 16.5 °C
From energy balance principal when the aluminium piece is heated and dropped in to the water , the final temperature of aluminium & the water becomes equal.
From the energy conservation principal
Heat lost from the aluminium piece = heat gain by the water
⇒
 (
( 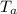 -
-  ) =
) = 
 (
(  -
-  ) ----------- (1)
) ----------- (1)
Put all the values in equation (1)
⇒ 101 × 0.9 × (100.1 - ) = 48.9 × 4.2 × (
) = 48.9 × 4.2 × (  - 16.5 )
- 16.5 )
⇒ 90.9 × (100.1 - ) = 205.38 × (
) = 205.38 × (  - 16.5 )
- 16.5 )
⇒ 100.1 - = 2.26 (
= 2.26 (  - 16.5 )
- 16.5 )
⇒ 100.1 - = 2.26
= 2.26  - 37.28
- 37.28
⇒ 3.26 = 137.38
= 137.38
⇒ = 42.14 °C
= 42.14 °C
Therefore the final temperature of the mixture is = 42.14 °C
Ответ:
Area of CMB = 12
Area of ABC = 24
Step-by-step explanation:
IDK how to prove it exactly but using logic I will go through the steps to findign the answer.
if DM is the midpoint of BC, it splits BC in half which also splites the area of BMC into two equal parts.
If the area of BMD = 6 and CMB = BMD * 2 then CMB = 6 * 2 = 12
Area of CMB = 12
If BM is the midpoint of AC, it splits AC in half which also splits the area into two equal parts.
If the area of CMB = 12 and ABC = CMB * 2 then ABC = 12 * 2 = 24
Area of ABC = 24