A 19.45 gram sample of copper is heated in the presence of excess iodine. A metal iodide is formed with a mass of 58.30 g. Determine the empirical formula of the metal iodide.
Solved
Show answers
More tips
- O Other What happens if you get scared half to death twice?...
- F Family and Home What s That Noise When a Kettle Boils? The Science of Water and Steam...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- W Work and Career How much does an honest traffic police officer earn in a day?...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- S Style and Beauty How to Get Rid of a Bruise: Tips and Tricks...
- H Health and Medicine Is Massage Necessary? Facts and Opinions...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet Best Antivirus: How to Choose Protection for Your Computer?...
- F Food and Cooking When is Easter in 2011?...
Answers on questions: Chemistry
- C Chemistry I need help in this problem for an assignment. Please...
- C Chemistry 2. Why is the salt solution heated...
- C Chemistry Explain why the definition of philosophy as the love of wisdom in insufficient?...
- C Chemistry What is 40% hydrogen 40% oxygen 20% magnesium ratio?...
- C Chemistry Which of the following is evidence that a chemical change is occurring? a. Cherry drink mix is added to water and the resulting solution is red. b. A white powder...
- C Chemistry I need help in this problem for an assignment. Please...
- C Chemistry How many atoms are in 0.30 moles of sulfur dioxide gas at stp? a. 6.0 x 10 ^23b. 1.806 x 10 ^23c. 2.01 x 10 ^24 d. 1,806...
- M Mathematics Write an equation of a polynomial in factored form with zeros at 3, -2, and 1...
- P Physics A 0.150-kg baseball moving at a speed of 45.0 m/s crosses the plate and strikes the 0.250-kg catcher s mitt (originally at rest). The catcher s mitt immediately...
- M Mathematics Um please answer with the right answer also follow my insta trying to get 1000 followers detsgt.andreszambrano ^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^ ill...

Ответ:
The empirical formula for the given compound is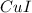
Explanation : Given,
Mass of Cu= 19.45 g
Mass of metal iodide = 58.30 g
Now we have to calculate the mass of iodide.
Mass of I = Mass of metal iodide - Mass of Cu
Mass of I = 58.30 - 19.45
Mass of I = 38.85 g
To formulate the empirical formula, we need to follow some steps:
Step 1: Converting the given masses into moles.
Moles of Cu =
Moles of I =
Step 2: Calculating the mole ratio of the given elements.
For the mole ratio, we divide each value of the moles by the smallest number of moles calculated which is 0.0306 moles.
For Cu =
For I =
Step 3: Taking the mole ratio as their subscripts.
The ratio of Cu : I = 1 : 1
Hence, the empirical formula for the given compound is
Ответ:
Most steps were skipped they couldn't predict what it would be they couldn't really observe I font think they used very many.