danielobanoyen
19.09.2020 •
Chemistry
A 237g sample of molybdnum metal is heated to 100.1 0C and then dropped into an insulated cup containing 244 g of water at 10.0 0C. If the final temperature of the water and metal in the cup is 15.3 0C, then what is the specific heat of molybdenum?
Solved
Show answers
More tips
- D Dating, Love, Relationships How to Overcome Jealousy: Tips and Tricks...
- H Health and Medicine Headache: A Comprehensive Guide to Treatment...
- G Goods and services Stock center - a modern way of optimizing logistics...
- F Food and Cooking How to Properly Wash a Down Jacket? Tips from Experts...
- C Computers and Internet Thin Client: What It Is and Why You Need It?...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
Answers on questions: Chemistry
- C Chemistry What are parts in the human stomach...
- C Chemistry In gluconeogenesis, ATP is utilized in the pyruvate carboxylase reaction and GTP is utilized in the PEP carboxykinase reaction. These are required, respectively,...
- C Chemistry Are the atoms of gold and carbon always moving or always motionless? always moving Or always motionless...
- C Chemistry A man ran 60.5 km, how far is this in yards?...
- B Business 1. The movie examines how Ray Kroc and the McDonald brothers create a fast food empire through entrepreneurship. Explain, using details from the film, how they were...
- M Mathematics Am i this right?? this is graded and no links...
- B Biology I need 10 important vocabulary words for rocks and minerals...
- M Mathematics Each dozen donuts sells for $6.99. What is the difference, in dollars, between the total sales from the new box over 6 months this year and the total sales from...
- M Mathematics Jackie ran 7 more miles than 2 times the number of miles Aidan ran this week. Jackie ran 39 miles this week. Part A: Write an equation that represents this situation,...
- M Mathematics ASAP Please (this is so i can post it)...

Ответ:
The specific heat of molybdenum is 0.254 joules per gram-Celsius.
Explanation:
We consider the system formed by the molybdenum metal and water as our system, a control mass inside an insulated cup, that is, a container that avoids any energy and mass interactions between system and surroundings.
From statement we notice that metal is cooled down whereas water is heated. According to the First Law of Thermodynamics, we know that:
Where:
Now we expand this identity by definition of sensible heat:
The specific heat of the metal is cleared within equation above:
If we know that ,
, 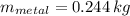 ,
, 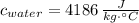 ,
, 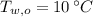 ,
, 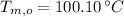 and
and 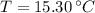 , the specific heat of molybdenum is:
, the specific heat of molybdenum is:
The specific heat of molybdenum is 0.254 joules per gram-Celsius.
Ответ:
2. True
3. True
Hope this helped! :)