stephensandrew4293
22.11.2020 •
Chemistry
A 50.0 mL sample of HCl required 25.81 mL of 0.1250 M KOH for neutralization. What is the molarity of the acid?
Solved
Show answers
More tips
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- C Computers and Internet How to Learn to Type Fast?...
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- D Dating, Love, Relationships How to Overcome Jealousy: Tips and Tricks...
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- W Work and Career How to Start Your Own Business: Tips and Recommendations...
- F Food and Cooking How to Make Delicious Cabbage Pies: The Best Recipes!...
- F Food and Cooking Discover Delicious Recipes You Can Make with Ground Meat...
Answers on questions: Chemistry
- C Chemistry Answer to this question?...
- C Chemistry 2.How many moles of Br2 are used when 14.00 moles of KBr are produced 3. If 102.3 grams of Fe(OH)3 are used, how many moles of H2O will be produced. 4.How many molecules of Hydrogen...
- C Chemistry Calculate the mass of water produced and enter in the data table above. Using the Law of Conservation of Matter, Provide a reason explaining the claim and evidence above by showing...
- C Chemistry Guys me with this problemjason,sarah and megan were discussing the kinetic particle theory.jason said that in a solid,the particles are close together.sarah said that all particles...
- C Chemistry How does photosynthesis use solar energy?...
- C Chemistry Ight this is for all em boy and girls out there who wanna be in an actual loving relationship. Not for the ones who just wanna get down, cuz i am sick and tired of boys and girls...
- C Chemistry Which of the following is an example of physical change?...
- C Chemistry Of the atoms below, __________ is the most electronegative. PLZ HELP I LL AWARD BRINLIEST....
- C Chemistry 13. Explain two of the methods of separating mixtures that are used to make wheat flour. Be specific about how each method is used. (14) Method 1: Method 2:...
- C Chemistry Any chemical or physical change that absorbs energy 1. cannot occur spontaneously. 2. forms products with less mass than the initial reactants. 3. is an endothermic process. 4. has...

Ответ:
0.065M
Explanation:
The reaction expression is given as:
HCl + KOH → KCl + H₂O
Given parameters:
Volume of KOH = 25.81mL = 0.02581L
Molarity of KOH = 0.125M
Volume of HCl = 50mL = 0.05L
Unknown:
Molarity of the acid = ?
Solution:
To find the molarity of the acid, HCl, let us use the mole concept.
First, find the number of moles of the base, KOH;
Number of moles of KOH = molarity of KOH x volume of KOH
Number of moles of KOH = 0.125 x 0.0258 = 0.003225mol
From the balanced reaction equation:
1 mole of KOH reacted with 1 mole of HCl
0.003225mol of KOH will react with 0.003255mole of HCl
Molarity of HCl =
Molarity of HCl =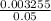 = 0.065M
= 0.065M
Ответ:
The mass of carbon dioxide formed in the reaction is 269 grams.
Explanation:
Law of conservation of mass states that mass can neither be created nor be destroyed but it can only be transformed from one form to another form.
This also means that total mass on the reactant side must be equal to the total mass on the product side.
The chemical equation for the decomposition of calcium carbonate follows:
Let the mass of carbon dioxide be 'x' grams
We are given:
Mass of calcium carbonate = 612 grams
Mass of CaO = 343 grams
Total mass on reactant side = 612 g
Total mass on product side = [343 + x] g
So, by applying law of conservation of mass, we get:
Hence, the mass of carbon dioxide formed in the reaction is 269 grams.