natalie2sheffield
05.03.2020 •
Chemistry
A certain mass of carbon reacts with 13.6 g of oxygen to form carbon monoxide. grams of oxygen would react with that same mass of carbon to form carbon dioxide, according to the law of multiple proportions.
Solved
Show answers
More tips
Answers on questions: Chemistry
- C Chemistry CH 9 Lecture Notes Predict the molecular shape for each of the following molecules/ions (a-g) from your CH 9 lecture slides. **Use the VSEPR model to predict the geometries...
- C Chemistry 2. What would charge would you expect a magnesium atom to form? O 2+ 017 2...
- C Chemistry 7. Which of the following properties identifies a pure substance? A. soluble in waterB. tasteless and odorlessC. has a constant boiling temperatureD. can be changed...
- C Chemistry A pure gold ring with a volume of 1.57 cm3 is initially at 11.4 oC. When it is put on someone s finger, it warms to 85.1 oF. How much heat does the ring absorb? (Specific...
- C Chemistry According to the law of the conservation of matter if you dissolve 25 g of sugar into 150 g of water then make sure should weigh how much...
- C Chemistry Choose the option that best completes the sentence. A scientific offers a scientific explanation about why something happens and is supported by a large number of experiments....
- C Chemistry Aplot of velocity versus substrate concentration for a simple enzyme-catalyzed reaction produces a this indicates that at some point, the enzyme is group of answer choices...
- C Chemistry Q4: the average heat evolved by the oxidation of food in an average adult per hour per kilogram of body weight is 7.20 ୩ ୩ ୦୰. assume the weight of an average adult...
- C Chemistry What is the ph of h3o+ = 2.4(10^-3) m? a.) 3 b.) 2.6 c.) 5.4 d.) -3...
- C Chemistry Hello, everyone! here is the problem i am stuck with.: based on the “national ambient air quality objectives”, the acceptable hourly average concentration of carbon...

Ответ:
The mass of oxygen that will react with same amount of carbon as in carbon monoxide is 23.31 grams.
Explanation:
Law of multiple proportions states that when two elements combine to form two or more compounds in more than one proportion. The mass of one element that combine with a given mass of the other element are present in the ratios of small whole number. For Example: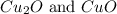
We are given:
Mass of oxygen in CO = 13.6 grams
For carbon monoxide (CO):Applying unitary method:
16 grams of oxygen are reacting with 12 grams of carbon
So, 13.6 grams of oxygen will be reacting with =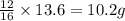 of carbon
of carbon
For carbon dioxideApplying unitary method:
12 grams of carbon are reacting with 32 grams of oxygen
So, 10.2 grams of carbon will be reacting with =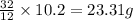 of oxygen
of oxygen
Hence, the mass of oxygen that will react with same amount of carbon as in carbon monoxide is 23.31 grams.
Ответ: