olivya2005d
26.02.2020 •
Chemistry
A concentration cell consists of two Sn/Sn2+ half-cells. The cell has a potential of 0.10 V at 25 ºC. What is the ratio of the Sn2+ concentrations in the two half-cells?
Solved
Show answers
More tips
- C Computers and Internet Dynamically Assigned IP Address: What Is It and How Does It Work?...
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- C Computers and Internet How to Learn to Type Fast?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- S Style and Beauty How to braid friendship bracelets?...
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking Which Calamari Salad is the Most Delicious?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- H Health and Medicine What is Autism? Understanding the Basics of This Neurodevelopmental Disorder...
Answers on questions: Chemistry
- C Chemistry Calculate the hydroxyl ion concentrationin 0.02m solution of h2so4...
- C Chemistry Enter a balanced equation for the combustion of ethanol....
- C Chemistry Condensation is when a liquid changes to a gas. true false...
- C Chemistry N If 10 mol of HCl are used, how many mol of H20 are formed? (use the balanced equation above) 3 1 10...
- C Chemistry Particles of a liquid a. are tightly packed together and stay in a fixed position. b. have no viscosity. c. decrease in volume with increasing temperature. d. are free...
- C Chemistry What makes the tea with milk and lemon curdle?...
- C Chemistry Agcl is soluble or insoluble? how do i identify this? ( by using table: solubility rules of salts in water)...
- M Mathematics Answer? What’s the answer I’m not sure...
- H History Why would the world be concerned that both India and Pakistan have nuclear weapons?...
- M Mathematics Find the volume of the sphere. Round to the nearest tenth....

Ответ:
The ratio of![\frac{[Sn^{2+}_{(diluted)}}{[Sn^{2+}_{(concentrated)}]}](/tpl/images/0524/9528/81ffa.png) in the cell is
in the cell is 
Explanation:
The half reactions for the cell is:
Oxidation half reaction (anode):
Reduction half reaction (cathode):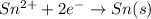
In this case, the cathode and anode both are same. So, will be equal to zero.
will be equal to zero.
To calculate cell potential of the cell, we use the equation given by Nernst, which is:
where,
n = number of electrons in oxidation-reduction reaction = 2
Putting values in above equation, we get:
Hence, the ratio of![\frac{[Sn^{2+}_{(diluted)}}{[Sn^{2+}_{(concentrated)}]}](/tpl/images/0524/9528/81ffa.png) in the cell is
in the cell is 
Ответ:
54 g
Explanation:
1 mole of water = H2O
mass of 1 mole of H2O= mass of h2 + mass of o
= 2× mass of h +mass of o
= 2×1+16 =18 g
1 mole of water = 18g
3moles of water = 18×3g= 54g