melissareid65
20.05.2021 •
Chemistry
a deep underground cavern contains 2.24 x 10^6 L of methane (CH4) gas at a pressure of 15.0 atm and a temperature of 42 C. How many moles of methane does this natural gas deposit contain?
Solved
Show answers
More tips
- A Animals and plants Want a Perfect Lawn? Learn How to Plant Grass the Right Way...
- A Animals and plants How to Properly Care for a Pet Decorative Rabbit at Home?...
- C Computers and Internet How to Check the Speed of My Internet?...
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
Answers on questions: Chemistry
- C Chemistry Nitric acid is a strong acid, sodium hydroxide is a strong base, and sodium nitrate is a soluble salt. Which of the following is the net ionic equation for the...
- C Chemistry In which of the following compounds does the carbonyl stretch in the IR spectrum occur at the lowest wavenumber? a. Cyclohexanone b. Ethyl Acetate c. λ- butyrolactone...
- C Chemistry Identify the goal of any scientific method...
- C Chemistry A pressure cooker allows food to be cooked under high pressure. Why is this useful?...
- C Chemistry Is blowing a dandelion a physical or chemical change? A. Physical B. Chemical...
- C Chemistry After three half lives what fraction of a radioactive sample has decayed...
- C Chemistry 2. Reaction between magnesium & sulphur...
- C Chemistry Which of the following ligands is not capable of exhibiting linkage isomerism? a. NCO- b. -OH c. -CN d. -SCN...
- C Chemistry Which of the following pairs are linkage isomers? A. [Co(NH3)4] [PtCl4] and [Pt(NH3)4] [CoCl4] B. [PdCl2(NH3)4]Br2 and [PdBr2(NH3)4]Cl2 C. [Cu(NH3)5NO3]SO4 and...
- C Chemistry A complex ion that forms in solution has a structure that:. a. can be determined simply by stoichiometry. b. can be predicted on the basis of electrical charge....

Ответ:
This natural gas deposit contains 1,300,813.008 moles.
Explanation:
An ideal gas is a theoretical gas that is considered to be composed of point particles that move randomly and do not interact with each other. Gases in general are ideal when they are at high temperatures and low pressures.
An ideal gas is characterized by three state variables: absolute pressure (P), volume (V), and absolute temperature (T). The relationship between them constitutes the ideal gas law, an equation that relates the three variables if the amount of substance, number of moles n, remains constant and where R is the molar constant of the gases:
P * V = n * R * T
In this case:
P= 15 atmV=2.24*10⁶ Ln= ?R= 0.082Replacing:
15 atm* 2.24*10⁶ L= n*0.082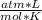 * 315 K
* 315 K
Solving:
n=1,300,813.008 moles
This natural gas deposit contains 1,300,813.008 moles.
Ответ:
KE = 1/2mv^2 = (.5)(.00420kg) (965m/s)^2 = 1960J = 1.9 x 10^3J
Explanation: