A gas container has an initial temperature of 348 K with an unknown pressure. When the temperature changes to 506 K the pressure is found to be 1.55 atm. What was the initial pressure in atm?
Solved
Show answers
More tips
- F Food and Cooking Discover How to Properly Prepare Dough for Rasstegai...
- P Philosophy Unidentified Flying Object - What is the Nature of this Phenomenon?...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
Answers on questions: Chemistry
- C Chemistry The amount of space that a gas takes up is its a. volume. b. mass. c. pressure. d. density....
- E English According to patel what role does government play in the current food system? explain whether the governments role is effective...
- B Biology Marine debris is best described as being composed of . a. solid garbage b. untreated sewage c. toxic chemicals d. things discarded by humans Please select the best answer from...
- M Mathematics Describe the difference between0.008 X 100 and 0.008 X 102....

Ответ:
1.06 atm
Explanation:
We are given the following variables to work with:
Initial pressure (P1): unknown
Initial temperature (T1): 348 K
Final temperature (T2): 506 K
Final pressure (P2): 1.55 atm
We are asked to find the initial pressure (P1). We want to choose a gas law equation that relates initial pressure and temperature to final pressure and temperature. Gay-Lussac's law does this:
We can rearrange the law algebraically to solve for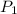 .
.
Substitute your known variables and solve:
Ответ:
decomposition is the answer it helped also explanation of it is below this answer
Explanation:
In a decomposition reaction, one reactant breaks down into two or more products. This is the reverse of a synthesis reaction. Replacement reactions occur when elements switch places in compounds