laureleiolson
13.01.2021 •
Chemistry
A radioactive substance has a half-life of 5 million years. What is the age of a rock in which 25% of the original radioactive atoms remain
Solved
Show answers
More tips
- H Health and Medicine Is it true that working with a computer is harmful to your eyesight?...
- S Sport When is the Champions League final?...
- H Health and Medicine Is Folic Acid a Necessary Vitamin?...
- W Work and Career How to Start Your Own Business: Tips and Recommendations...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Chemistry
- C Chemistry Group the labels according to the process they describe....
- C Chemistry Which form of technology had to be invented before these large moons were observed...
- C Chemistry I need help with this question please....
- C Chemistry Which practice may be considered unethical To some but prevents unnecessary human harm...
- C Chemistry How can atoms have more than 32 electons?...
- C Chemistry Gas y effuses half as fast as o2. what is the molar mass of gas y?...
- C Chemistry Draw a six-carbon alkyne that can exist as diastereomers. (the molecule should contain only carbon and hydrogen atoms.)...
- C Chemistry Both of the silicon-chlorine single bonds in sicl4 are polar. in which direction should the polarity arrows point?...
- C Chemistry Which group iia metal magnesium or calcium is more active cite your evidence?...
- C Chemistry How many unpaired electrons are in the beryllium atom?...

Ответ:
The given information gives us the clue that deuterium and tritium react together. We need to find the balanced nuclear reaction of deuterium and tritium; however, first, let me explain you what deuterium and tritium are.
Deuterium and tritium are two isotopes of hydrogen having atomic mass of 2 and 3 respectively. Deuterium is represented by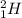 , whereas tritium is written as
, whereas tritium is written as 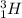 .
.
When deuterium and tritium collide together, they form Helium atom, which is written as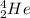 , and also release energy (and shoot out one neutron).
, and also release energy (and shoot out one neutron).
Hence, the balanced nuclear reaction will be the following:
-i