trying2passs
12.03.2020 •
Chemistry
A reaction was performed in which 3.6 g 3.6 g of benzoic acid was reacted with excess methanol to make 1.3 g 1.3 g of methyl benzoate. Calculate the theoretical yield and percent yield for this reaction.
Solved
Show answers
More tips
- F Food and Cooking Kick Your Taste Buds Into Gear with This Authentic and Delicious Recipe for Solyanka...
- O Other What happens if you get scared half to death twice?...
- F Family and Home What s That Noise When a Kettle Boils? The Science of Water and Steam...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- W Work and Career How much does an honest traffic police officer earn in a day?...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- S Style and Beauty How to Get Rid of a Bruise: Tips and Tricks...
- H Health and Medicine Is Massage Necessary? Facts and Opinions...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet Best Antivirus: How to Choose Protection for Your Computer?...
Answers on questions: Chemistry
- C Chemistry How can volatility be tested in a laboratory...
- C Chemistry What two features must a sample have if it is to accurately represent a population...
- C Chemistry Choose the largest atom from each ofthe following groups. Explain your answer.a) O , N , C...
- C Chemistry The heat of reaction for a chemical reaction can be calculated by finding the sum of the bond energies of the products and subtracting that from the sum of the bond energies...
- C Chemistry The unstable nucleus that breaks apart is called A) Daughter Nucleus B) Parent Nucleus C) Beta Particle D) Alpha Particle...
- C Chemistry The enzyme urease enhaces the rate of urea hydrolysis at pH8.0 and 20 degree of centigrade by a factor of 1014. If a given quantity of urea in 5 min at pH 8.0 and 20 degree...
- C Chemistry In general how are word equations written to describe chemical equations...
- C Chemistry What does the number 84 in the name krypton-84 represent? b. atomic number c. mass number d. twice the number of protons...
- C Chemistry The element, boron, is represented by which of the following chemical symbols? be br b ba...
- M Mathematics If two angles are supplementary the their sum is 180...

Ответ:
The percent yield of the reaction is 32.34 %
Explanation:
To calculate the number of moles, we use the equation:
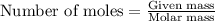 .....(1)
.....(1)
For benzoic acid:Given mass of benzoic acid = 3.6 g
Molar mass of benzoic acid = 122.12 g/mol
Putting values in equation 1, we get:
The chemical equation for the reaction of benzoic acid and methanol is:
By Stoichiometry of the reaction
1 mole of benzoic acid produces 1 mole of methyl benzoate
So, 0.0295 moles of benzoic acid will produce =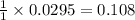 moles of methyl benzoate
moles of methyl benzoate
Now, calculating the mass of methyl benzoate from equation 1, we get:Molar mass of methyl benzoate = 136.15 g/mol
Moles of methyl benzoate = 0.0295 moles
Putting values in equation 1, we get:
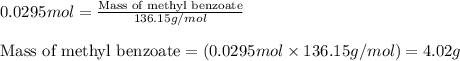
To calculate the percentage yield of methyl benzoate, we use the equation:Experimental yield of methyl benzoate = 1.3 g
Theoretical yield of methyl benzoate = 4.02 g
Putting values in above equation, we get:
Hence, the percent yield of the reaction is 32.34 %
Ответ:
i don't really know what you are trying to say here, but what i think you are saying is what sounds like a more impressive job?? in my opinion, pseudoscience sounds more fancy and interesting. Since not a lot of people really knows what that is, it sparks conversation.