vperez5765
27.07.2021 •
Chemistry
A sample of aluminum, which has a specific heat capacity of , is put into a calorimeter (see sketch at right) that contains of water. The temperature of the water starts off at . When the temperature of the water stops changing it's . The pressure remains constant at .Calculate the initial temperature of the aluminum sample. Be sure your answer is rounded to significant digits.
Solved
Show answers
More tips
- H Health and Medicine How to Whiten Teeth and Get the Perfect Smile...
- F Food and Cooking How to Make Thin Pancakes: Recipe and Tips...
- S Style and Beauty Is Hot Scissor Haircutting Beneficial or Dangerous?...
- S Style and Beauty How to Get Rid of Under Eye Bruises?...
- F Food and Cooking Is Bacon Good for You?...
- S Style and Beauty Discover the Art of Nail Design: How Do You Paint Your Nails?...
- P Philosophy How to Develop Extrasensory Abilities?...
- O Other Everything You Need to Know About Kudyabliks...
- C Computers and Internet The Twitter Phenomenon: What it is and How to Use it...
- C Computers and Internet How to Choose a Laptop: Expert Guide and Tips...
Answers on questions: Chemistry
- S Social Studies Bodies A and B have equal mass. Body B is initially at rest. Body A collides with body B in a one–dimensional elastic collision. Which statement is true?...
- H History How did the crusades affect the european social structure and trade? ?...
- M Mathematics Robin, Pete, and Jason each wrote expressions to represent their hourly earnings for an after-school job where h represents the number of hours worked. Robin: 9.50h + 16...

Ответ:
Complete Question
A sample of aluminum, which has a specific heat capacity of 0.897 JB loc ! is put into a calorimeter (see sketch at right) that contains 200.0 g of water. The aluminum sample starts off at 85.6 °C and the temperature of the water starts off at 16.0 °C. When the temperature of the water stops changing it's 20.1 °C. The pressure remains constant at 1 atm. Calculate the mass of the aluminum sample.
Explanation:
From the question we are told that:
Heat Capacity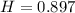
Mass of water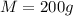
Initial Temperature of Aluminium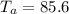
Initial Temperature of Water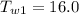
Final Temperature of Water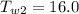
Generally
Heat loss=Heat Gain
Therefore
Ответ: