skinniestoflegends
03.06.2020 •
Chemistry
A sample of gas at 25 C has a volume of 11 L and exerts a pressure of 0.868 atm. How many moles of gas are in the sample?
0.39 mol
3.9 mol
9.3 mol
87 mol
Solved
Show answers
More tips
- S Style and Beauty What to Do When Hair Starts Falling Out?...
- H Health and Medicine Novomin: What is it and how to use it?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
Answers on questions: Chemistry
- C Chemistry An oxide of phosphorus contains 56.4% phosphorus and 43.6% oxygen. it s relative molecular mass is 220. find both the empirical and the molecular formula of the oxide...
- C Chemistry Describe the solubility of al(oh3 with respect to ph....
- C Chemistry What term refers to elements in group 18 that only react with other elements under special laboratory conditions? noble gases metalloid nonmetals halogen...
- C Chemistry How many cubic feet of air at standard conditions 1.00 atm are required to inflate a bicycle tote of .50 cu. ft. to a pressure of 3.00 atmospheres?...
- C Chemistry How did ernest ruth ford s discovery affect the model of the atom?...
- C Chemistry I need help A.S.P please, it’s a test...
- S Social Studies Critical Thinking Question Which of the disadvantages of a Command Economy is shown in these panels....
- M Mathematics Given the figure below, find the values of x and z...
- E English IF YOU WANT TO PLAY K A H O O T THE JOIN CODE IS 9568888 PLEASE JOIN. THE QUESTION WILL SHOW UP ON YOUR SCREEN TOOO....
- M Mathematics The products of respiration are the of photosynthesis. A.) products B.) reactants C.) powerhouses...

Ответ:
0.39 moles of gas are in the sample
Explanation:
An ideal gas is a theoretical gas that is considered to be composed of point particles that move randomly and do not interact with each other. Gases in general are ideal when they are at high temperatures and low pressures.
The pressure, P, the temperature, T, and the volume, V, of an ideal gas, are related by a simple formula called the ideal gas law:
P*V = n*R*T
where P is the gas pressure, V is the volume that occupies, T is its temperature, R is the ideal gas constant, and n is the number of moles of the gas.
In this case:
P= 0.868 atmV= 11 Ln= ?R= 0.082Replacing:
0.868 atm*11 L= n*0.082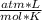 *298 K
*298 K
Solving:
n= 0.39 moles
0.39 moles of gas are in the sample
Ответ: