A sample of krypton gas at a pressure of 1.02 atm and a temperature of 167 C, occupies a volume of 587mL. If the gas is heated at constant pressure until its volume is 849 mL, the temperature of the gas sample will be?
Solved
Show answers
More tips
- F Food and Cooking How to Make Thin Pancakes: Recipe and Tips...
- S Style and Beauty Is Hot Scissor Haircutting Beneficial or Dangerous?...
- S Style and Beauty How to Get Rid of Under Eye Bruises?...
- F Food and Cooking Is Bacon Good for You?...
- S Style and Beauty Discover the Art of Nail Design: How Do You Paint Your Nails?...
- P Philosophy How to Develop Extrasensory Abilities?...
- O Other Everything You Need to Know About Kudyabliks...
- C Computers and Internet The Twitter Phenomenon: What it is and How to Use it...
- C Computers and Internet How to Choose a Laptop: Expert Guide and Tips...
- C Computers and Internet How to Choose a Monitor?...
Answers on questions: Chemistry
- S Social Studies Why is 1 divided by 50 an equivalent ratio to 2 divided by 100?...
- A Advanced Placement (AP) Multiple Choice Knowing the number of species in existence assists scientists in determining the species that are in danger of extinction. O True O False...
- B Biology Use the diagram below to match the letter with the name of the layer. 1. inner core2. outer core3. mantle4. crust...
- A Arts What is the form used most in pop songs? A. Verse and chorus B. Ternary form C. Rondo form D. Call and response...

Ответ:
Three atomic orbitals are related to a p orbital.
Further Explanation:
Quantum numbers:
Quantum numbers govern the size, energy, shape, and orientation of an orbital. The four quantum numbers are as follows:
1. Principal Quantum Number (n): It denotes the principle electron shell. The values of n are positive integers (1, 2, 3,…).
2. Angular Momentum Quantum Number (l): It represents the shape of an orbital. The value of l is an integer from 0 to (n-1). The value of l for s orbital is 0, that for p orbital is 1, for d orbital it is 2 and for f orbital, it is 3. (Refer to the table in the attached image).
3. Magnetic Quantum Number : This quantum number represents the orientation of the orbital in space. The value of
: This quantum number represents the orientation of the orbital in space. The value of  lies between –l to +l. The formula to calculate the value of
lies between –l to +l. The formula to calculate the value of  is as follows:
is as follows:
Therefore, the total number of values for a given value of l is 2l+1.
values for a given value of l is 2l+1.
4. Electron Spin Quantum Number : It represents the direction of the electron spin. Its value can be
: It represents the direction of the electron spin. Its value can be  or
or  .
.
As we know the value of l for p orbital is 1.
The total value of can be calculated as follows:
can be calculated as follows:
So a p sublevel has three atomic orbitals. These are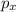 ,
,  and
and  .
.
Learn more:
1. Allowed values of :
2. Calculation of volume of gas:
Answer details:
Grade: Senior School
Subject: Chemistry
Chapter: Structure of the atom
Keywords: quantum numbers, n, l, ml, ms, 1, 2, 3, 0, p orbital, three atomic orbitals.