A teacher has given a lab student a white chemical sample and asks her to confirm that it contains 7.03 × 1023 formula units of sodium hydroxide (NaOH). Using the balance in the laboratory, what mass of NaOH would the student have to find to verify that her teacher was correct? Round your answer to 3 significant figures.
Solved
Show answers
More tips
- A Auto and Moto Experience the World of the Most Expensive Cars on the Planet...
- S Style and Beauty How to Get Rid of a Double Chin?...
- F Food and Cooking How to Cook Julienne? Recipes and Tips...
- D Dating, Love, Relationships 10 Useful Tips on How to Survive a Breakup?...
- F Food and Cooking Apple Cider Vinegar: The Ultimate Health and Beauty Solution...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
Answers on questions: Chemistry
- C Chemistry worth 100 pointsthis chart shows four atoms, labeled w, x, y, and z. these atoms can combine with each other to form molecules.which atoms could join together to form...
- C Chemistry Why is that helium is shown here with the group ii alkali earth metals...
- M Mathematics What is the value of x? enter your answer in the box. x =...
- M Mathematics The difference of eight times a number and four times the same number...
- H History Describe briefly the articles of confederation- what were their weaknesses? what was the intention of the authors when creating the articles?...
- H History If you were to stand on the alligators tail vertically , what might it resemble...
- E English Which answer here is the definition for this word or phrase? sacred writings scripturevariantdiction...
- M Mathematics Use the binomial theorem to expand (2a + 2b)5. a. 32a5 + 80a4b + 320a3b2 + 320a2b3 + 80ab4 + 10b5 b. 32a5 + 160a4b + 320a3b2 + 320a2b3 + 160ab4 + 32b5 c. 32a5 + 160a4b...
- M Mathematics Make n the subject of the formula m=7n...
- H Health Which of these is most often associated with an eating disorder? oa. depressionob. happinessinfectionfitness...

Ответ:
What is the mole fraction of a solution that contains 200 g of magnesium hydroxide in 225 g of water
0.215
Explanation:
Given parameters:
Mass of magnesium hydroxide = 200g
Mass of water = 225g
Unknown:
Mole fraction of solution = ?
Solution:
The mole fraction is a measurement of concentration that expresses the number of moles of solute to that of solution.
Mole fraction =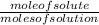
A solution is made up of solute and solvent
Number of moles =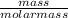
Molar mass of Mg(OH)₂ = 24.3 + 2(16 + 1) = 58.3g/mol
Molar mass of H₂O = 2(1) + 16 = 18g/mol
Number of moles of Mg(OH)₂ =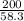 = 3.43moles solute
= 3.43moles solute
Number of moles of H₂O =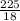 = 12.5moles solvent
= 12.5moles solvent
Mole fraction =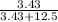 = 0.215
= 0.215