lexieforlife911
28.06.2019 •
Chemistry
A. what is the mass number of the particle emitted from the nucleus during beta minus (β–) decay? b. what kind of charge does the particle emitted from the nucleus during beta minus (β–) decay have? c. what is another name for a beta minus (β–) particle?
Solved
Show answers
More tips
- S Style and Beauty Lamination of Hair: How it Works and What it is?...
- C Computers and Internet Best Applications for Your iPad: Review of the Best Candidates for Installation...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
Answers on questions: Chemistry
- S Social Studies Tell me some questions you may have about sexual harassment...
- B Biology Wait what year is it???...
- M Mathematics CAN ANYONE PLEASE HELP THANKS...
- M Mathematics The school band sold t-shirts to raise money for an upcoming trip. the band sold 400 t-shirts on saturday. on sunday, it will sell 30 percent of that amount. which statements...

Ответ:
For a. Mass number for the particle is 0.
For b. The charge on the particle is -1.
For c. Another name for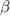 - particle is electron.
- particle is electron.
Explanation:
Beta- minus decay is the decay process in which beta-particle is emitted. The mass number of the nuclei remains same and the mass number of the particle is 0. The charge on the particle is -1 and the emitted particle is also called as electron.
For a. The mass number for the particle is 0.
For b. The charge on the particle is -1.
For c. Another name for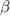 - particle is electron.
- particle is electron.
Ответ:
0.51
Explanation:
Given the Nernst equation;
E= E° - 0.0592/n logQ
E= 355 mV or 0.355 V
E° = 0.34 - 0= 0.34 V
n= 2(two electrons were transferred in the process)
Equation of the reaction;
H2(g) + Cu^2+(aq) > 2H^+(aq) + Cu(s)
Substituting values;
0.355 = 0.34 - 0.0592/2 log([H^+]/1)
0.355 - 0.34 = - 0.0296 log [H^+]
0.015/-0.0296 = log [H^+]
Antilog (-0.5068) = [H^+]
[H^+] = 0.311 M
pH = -log[H^+]
pH= - log(0.311 M)
pH = 0.51