A0.10 m solution of a weak monoprotic acid has a ph of 3.40 at 25°c. what is the acid-ionization
constant, ka, for this acid?
a) 1.6 x 10-6
b) 4.0 x 10-4
c) 3.4 x 10-5
d) 1.2 x 10-3
e) 1.8 x 10-7
Solved
Show answers
More tips
- A Art and Culture The History and Characteristics of Jazz Bands: A Deep Dive...
- W Work and Career Can Skill Alone Make You a Professional?...
- F Family and Home Daughter says: If you don t want to do it, don t do it. Should we persuade her?...
- S Science and Technology How to Secure Exam Sessions: Silence Mobile Phones in the Classroom...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- P Philosophy Is Everything We Strive for Eventually Achieved and Destroyed?...
- S Society and Politics Understanding Politics and Its Role in the Development of Civilization...
- P Philosophy Why Did God Create Man and Place Him in Obscurity?...
- S Society and Politics Skoptsy: Who They Are and How They Perform Castration?...
- O Other Childhood Fears: What Many of Us Experienced...
Answers on questions: Chemistry
- B Biology How do loggerhead turtles use Earth’s magnetic poles when they migrate?...
- C Computers and Technology The list method reverse reverses the elements in the list. define a function named reverse that reverses the elements in its list argument (without using the method...
- H History Most of the new settlers in south carolina were members of what group? o a. religious dissenters from northern colonies b. runaway slaves from other colonies o o c....
- B Biology create a pie chart showing the percentages of plastic that was recycled, burned or placed in landfills in 2015 (out of the total generated)...

Ответ:
The correct answer is A) 1.6 x 10-6
Explanation:
A weak monoprotic acid has the following dissociation equilibrium. At the beggining (t=0), the concentration of the monoprotic acid (HA) is equal to 0.10 M and the concentration of the ions H⁺ and A⁻ is zero (no dissociation). At a time t, dissociation occur and there is x concentration of H⁺ and A⁻ which is given by the dissociation constant Ka.
HA(aq) ⇄ H⁺(aq) + A⁻(aq)
t=0 0.10 M 0 0
t -x +x +x
eq 0.10 M-x x x
Ka=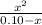
As the pH is 3.40, we can calculate the concentration of both H⁺ and A⁻, as follows:
pH= - log (conc H⁺)= -log x
⇒ x =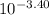 = 3.98 x 10⁻⁴
= 3.98 x 10⁻⁴
Now we introduce x in the previous equation to calculate Ka:
Ka=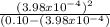
Ka= 1.59 x 10⁻⁶ ≅ 1.60 x 10⁻⁶
Ответ:
29.9 moles
2C²H¹⁰ needs 13 moles of O²
4.6 C⁴H¹⁰ needs X moles of O²
X= 13× 4.6 ÷ 2 = 59.8 ÷ 2 = 29.9 moles