ineedhelplolol
09.07.2019 •
Chemistry
A0.320 g sample of a carboxylic acid is burned in oxygen, producing 0.571 g of co2 and 0.235 g of h2o. determine the empirical formula of the carboxylic acid.
Solved
Show answers
More tips
- A Auto and Moto How to choose the right drive for your BMW...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
Answers on questions: Chemistry
- C Chemistry Chris is about to do an experiment to measure the density of water at several temperatures. his teacher has him prepare and sign a safety contract before beginning the experiment....
- C Chemistry 0.000786 written in scientific notation is?...
- C Chemistry 1. What happens to the amount of energy as you move up the energy pyramid. 2. Describe how the other organisms would be affected if the secondary consumer in a food chain were removed....
- C Chemistry What characteristics should a good scientific question have...
- C Chemistry Trapped gas within the sun is known as...
- C Chemistry Susan walks her dog around the neighborhood. she walks 3 blocks north, 5 blocks east, 2 blocks south, 3 blocks west, 1 block south, and 2 blocks west to end up back at her house....
- C Chemistry Ido not know how is my answered...
- C Chemistry Barium sulfate is used to make the digestive tract visible in X-rays. For it to be visible, it must remain insoluble. A pharmacist preparing a slurry of barium sulfate should choose...
- C Chemistry The Cell Map is a learning tool developed by Marcy Osgood and Karen Ocorr, the authors of the study guide companion for Lehninger. a. True b. False...
- C Chemistry Calculate the mass of chromium produced from 50g of Cr2O3-...

Ответ:
- Empirical formula of the carboxylic acid is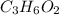 .
.
Solution:- When the acid is burned then all it's carbon goes to form carbon dioxide and all the hydrogen goes to form water.
So, we convert the grams of carbon dioxide to moles and calculate the moles of carbon from it as one mol of carbon dioxide contains one mol of carbon.
=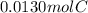
Similarly, we convert the grams of water to moles and these moles are used to calculate the moles of hydrogen as each mol of water contains two moles of hydrogen.
=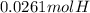
Now, we convert the moles of C and H to grams and subtract the sum of their mass from the given mass of the acid to calculate the mass of oxygen present in the acid.
= 0.156 g C
Similarly,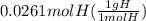
= 0.0261 g H
mass of oxygen in acid = 0.320 g - (0.156 g + 0.0261 g)
= 0.320 g - 0.1821 g
= 0.1379 g
Let's convert the grams of oxygen to moles;
= 0.00862 mol O
Next step is to calculate the mol ratio and for this we divide the moles ofch by the least one of them. We have least moles of oxygen, so let's divide the moles of each by the moles of oxygen.
Mol ratio sgould be whole number ratio, if it is not then we multiply all of them by 2, 3, 4 and so on to get the smallest whole number ratio. Here if we multiply by 2 then it works.
So, C = 2(1.5) = 3
H = 2(3.0) = 6
O = 2(1.0) = 2
Hence, the empirical formula of the carboxylic acid is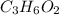 .
.
Ответ:
The coke is produced by the carbonization of coal.