A2.60 g sample of titanium metal chemically combines
withchlorine gas to form 10.31g of a titanium chloride.
a. what is the empirical formula of the titaniumchloride
b. what is the percent by mass of titanium and chlorine in
thesample?
Solved
Show answers
More tips
- S Science and Technology Exploring Our Galaxy: How Many Planets are in the Milky Way?...
- W Work and Career Can Skill Alone Make You a Professional?...
- C Computers and Internet How to Top Up Your Skype Account Without Losing Money?...
- P Philosophy Unidentified Flying Object - What is the Nature of this Phenomenon?...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
Answers on questions: Chemistry
- H History The Reconstruction plans of President Abraham Lincoln and President Andrew Johnson included a provision for the- resumption of full participation in Congress by...
- M Mathematics Lesson 5: line and angle relationships unit test connexus...
- M Mathematics A group of 7 students was asked, How many hours did you watch television last week? Here are their responses. 12, 5, 14, 6, 17, 7, 15 Send data to calculator Find...
- M Mathematics ANGLES question use the image attached below to help...

Ответ:
For a: The empirical formula for the given compound is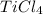
For b: The percent by mass of titanium and chlorine in the sample is 25.55 % and 74.78 % respectively.
Explanation:
For a:We are given:
Mass of Titanium = 2.60 g
Mass of sample = 10.31 g
Mass of Chlorine = 10.31 - 2.60 = 7.71 g
To formulate the empirical formula, we need to follow some steps:
Step 1: Converting the given masses into moles.Moles of titanium =
Moles of Chlorine =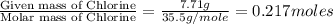
Step 2: Calculating the mole ratio of the given elements.For the mole ratio, we divide each value of the moles by the smallest number of moles calculated which is 0.054 moles.
For Titanium =
For Chlorine =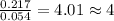
Step 3: Taking the mole ratio as their subscripts.
The ratio of Ti : Cl = 1 : 4
Hence, the empirical formula for the given compound is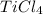
For b:To calculate the percentage by mass of substance in sample, we use the equation:
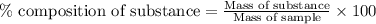 .......(1)
.......(1)
For Titanium:Mass of sample = 10.31 g
Mass of titanium = 2.60 g
Putting values in above equation, we get:

For Chlorine:Mass of sample = 10.31 g
Mass of chlorine = 7.71 g
Putting values in above equation, we get:
Hence, the percent by mass of titanium and chlorine in the sample is 25.55 % and 74.78 % respectively.
Ответ:
Its B, C, and D.
Look at photo for proof.