A25.00 ml sample of an aqueous solution of ba(oh)2 requires 18.45 ml of 0.01500 m hcl (aq) for its neutralization. what is the molarity of the ba(oh)2 solution?
Solved
Show answers
More tips
- C Computers and Internet Отправляем смс через интернет: легко и просто...
- H Health and Medicine Want to Lose Weight? Here s How Many Calories You Need Per Day...
- H Health and Medicine How Many Ribs Do Humans Have?...
- C Computers and Internet Dropbox: What is it and How to Use it...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- A Auto and Moto How many blood alcohol level units are allowed in Russian traffic laws?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- H Health and Medicine How to Deal with Heat Stroke?...
- H Health and Medicine Sunstroke: Causes, Symptoms, and Precautions...
Answers on questions: Chemistry
- C Chemistry The job outlook for physical therapists a) is weak to very poor. b) depends on the economy. c) will remain stable. d) will improve over time....
- C Chemistry Can someone me answer this question? ?...
- C Chemistry Adrug company is testing the effectiveness os a new blood pursur medicine using rats as the test subject. what are some possible factors that must stay current during...
- C Chemistry Whats the scientific notation of 0.00000923...
- C Chemistry Which equipment would you separate a liquid mixture by distillation? a freezer sand or gravel two flasks, a heat source, and a condensing tube a sieve, such as a colander...
- C Chemistry What is the scientfic notation of 0.00000923...
- C Chemistry Which of the following is a component of cell theory that is supported by the diagram...
- C Chemistry HEL PLEAS I AM GIVING BRAINLIEST PLEAS How many atoms are in 14.05 grams of silicon, Si? WITH EXPLANATION PLEASE...
- C Chemistry Fill in the chart below to identify and describe the functional groups associated with organic chemistry. Alcohol, aldehyde, ketone, fatty acid, ether...
- C Chemistry How Did the South American Plate and African Plate move I will mark as Brainliest and will give a lot of points...

Ответ:
5.54 x 10⁻³ M
Further explanationGiven:
Neutralization reaction between:
25.00 ml of Ba(OH)₂18.45 ml of 0.015 M HClQuestion:
What is the molarity of the Ba(OH)₂ solution?
The Process:
Let us say the molarity of the Ba (OH)₂ solution as x M.
Step-1: prepare moles for each reagent
Step-2: neutralization
We use the ICE table to see how neutralization occurs between acid and base.
Balanced reaction:
Initial: 25x 0.277 - -
Change: - ¹/₂ · (0.277) -0.277 +¹/₂ · (0.277) +0.277
Equlibrium: - - +¹/₂ · (0.277) +0.277
Neutralization causes no excess of hydrogen or hydroxide ions in solution. In the end, the number of acid and base reactions is balanced. In other words, the two reagents have run out with nothing left.HCl acts as a limiting reagent.Step-3: calculate the molarity of the Ba(OH)₂ solution.
We consider Ba (OH) from the initial, change, and equilibrium stages.
Thus, the molarity of the Ba(OH)₂ solution is 5.54 x 10⁻³ M.
_ _ _ _ _ _ _ _ _ _
Alternative Steps
Valence of base = the number of OH⁻ ionsValence of acid = the number of H⁺ ionsNeutralization: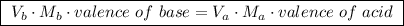
Thus the same results were obtained. The molarity of Ba (OH) ₂ solution is 5.54 x 10⁻³ M.
Learn moreWhat is the concentration of a solution formed by diluting 25.0 ml of a 3.2 M NaCl solution to 135.0 ml? link How much of these solutions he needs to drain and replace with 70% acid solution to obtain 100g of 60% acid solution? linkHow many liters of the 50% solution and how many liters of the 90% solution will be used? linkОтвет: