Aballoon filled with helium gas has a volume of 500 ml at a pressure of 1 atm. the balloon is released and reaches an altitude of 6.5 km, where the pressure is 0.5 atm. if the temperature has remained the same, what volume does the gas occupy at this height?
Solved
Show answers
More tips
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
- P Photography and Videography How to Choose the Perfect Photo Paper for Your Images?...
Answers on questions: Chemistry
- C Chemistry calculate the heat required to melt 25.7g of solid methanol ( at its melting point ( Hfus=3.22KJ/mol , Mm=32g/mol)...
- C Chemistry How to know if you can heat a laboratory flask and other glassware?...
- C Chemistry 100 POINTS!! PLEASE HELP WILL MARK BRAINLIEST...
- C Chemistry How many of the following numbers contain 3 significant figures? 0.509 1.050 0.0500 1.06 x 1024...
- C Chemistry When finding the mean, or average, of a set of numbers you divide the sum of the group of numbers by the number of items in the group. What is the average height...
- C Chemistry Can someone help me. Explain, stepwise, how DNA codes for a protein. Use a diagram to help draw or find and label one...
- C Chemistry How a change in DNA can lead to a change in phenotype. With an example...
- C Chemistry Which of the following is a pure substance made up of one type of atom? A) bronze B) lead C) steel D) brass...
- C Chemistry Net ionic equation for sodium carbonate and sulfuric acids?...
- C Chemistry A 0.530 M Ca(OH)2 solution was prepared by dissolving 36.0 grams of Ca(OH)2 in enough water. What is the total volume of the solution thus formed? (4 points)...

Ответ:
Volume of the gas occupy at this height will be 1 L.
Explanation:
Volume of the balloon when pressure of helium gas was 1 atm :
= 500 mL = 0.5 L =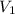 (1000 mL= 1 L)
(1000 mL= 1 L)
Volume of the balloon when the pressure of helium gas is 0.5 atm :
V=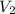
According to Boyle's Law:
Volume of the gas occupy at this height will be 1 L.
Ответ:
A. Fungus, Plants, and Animals
B. Bacteria, Archaea, and Eukarya
C. Protist, Fungus, and Plants
D. Bacteria, Virus, and Eukarya
i think it’s c :)