Officialavm
07.09.2019 •
Chemistry
Achemist wants to find kc for the following reaction at 751 k: 2nh3(g) + 3 i2 (g) latex: \longleftrightarrow ⟺ 6hi(g) + n2(g) kc = use the following data at 751 k to find the unknown kc: (1) n2(g) + 3 h2(g) latex: \longleftrightarrow ⟺ 2 nh3(g) kc1 = 0.282 (2) h2(g) + i2(g) latex: \longleftrightarrow ⟺ 2 hi(g) kc2 = 41 enter to 0 decimal places.
Solved
Show answers
More tips
- C Computers and Internet Make Money Online: Secrets and Essential Ways...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- C Computers and Internet How to Learn to Type Fast?...
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- D Dating, Love, Relationships How to Overcome Jealousy: Tips and Tricks...
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- W Work and Career How to Start Your Own Business: Tips and Recommendations...
- F Food and Cooking How to Make Delicious Cabbage Pies: The Best Recipes!...
Answers on questions: Chemistry
- C Chemistry What is the molarity of a solution of 3.2 moles of zinc chloride in 8.7 L of solution? Record your answer with one decimal place. Write the number only, no units....
- M Mathematics The mean is always one of the data values...
- B Biology Which statement correctly describes the bonding between water molecules?...
- S Social Studies . The Supreme Court takes on a case about the constitutional merits of a law. After hearing arguments for and against the law, they issue a verdict that the law goes...

Ответ:
The equilibrium constant for the total reaction is
Explanation:
We are given:
We are given two intermediate equations:
Equation 1:
The expression of for the above equation is:
for the above equation is:
Equation 2:
The expression of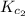 for the above equation is:
for the above equation is:
Cubing both the sides of equation 2, because we need 3 moles of HI in the main expression if equilibrium constant.
Now, dividing expression 1 by expression 2, we get:
The above expression is the expression for equilibrium constant of the total equation, which is:
Hence, the equilibrium constant for the total reaction is
Ответ:
Step-by-step explanation:
For this case we can model the problem as a cylinder.
The volume of a cylinder is given by:
Where,
Ab: base area
h: cylinder height
The volcano is 46 centimeters tall and has a
volume of 800 cubic centimeters.
Substituting values in the equation we have:
From here, we can clear the area of the base
An equation that can be used to find the area of the circular base is: