alejandra1201
19.09.2019 •
Chemistry
Acompound of formula xcl3 reacts with aqueous agno3 to yield solid agcl according to the following equation: xcl3(aq)+3agno3(aq)→x(no3)3(aq)+3agcl(s) when a solution containing 0.521 g of xcl3 was allowed to react with an excess of aqueous agno3, 1.68 g of solid agcl was formed. what is the identity of the atom x?
Solved
Show answers
More tips
- H Health and Medicine How to Treat Whooping Cough in Children?...
- L Leisure and Entertainment Unlocking the Secrets of Fast and Effective Tectonic Learning...
- A Animals and plants Уход за джунгариками: полезные советы и рекомендации...
- S Style and Beauty How to knit a hooded cowl?...
- S Style and Beauty How to Break in New Shoes: 7 Simple Methods...
- D Dating, Love, Relationships 10 Useful Tips on How to Survive a Breakup?...
- A Art and Culture How to Learn Screaming: Step-by-Step Guide for Beginners...
- A Art and Culture Attention, the Final Episode of Margo is Almost Here!...
- H Health and Medicine Novomin: What is it and how to use it?...
- L Leisure and Entertainment How to Land on the Moon: Your Comprehensive Guide...
Answers on questions: Chemistry
- G German More points ig lolllllllllllllllllllllll...
- S Social Studies What does the us government regulate?...
- B Biology What is the main purpose of Meiosis? To turn a diploid cell into 4 genetically different haploid cells. To turn a diploid cell into 2 copies that are genetically the same. To...
- M Mathematics Determine if the equation is parallel, perpendicular, or neither; y = -3/2x + 4 and y = 2/3x - 7...
- E English Compare and Constrast Dark they were golden eye short story and radio version...

Ответ:
Aluminum
Explanation:
Mass of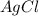 = 1.68 g
= 1.68 g
Molar mass of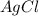 = 143.32 g/mol
= 143.32 g/mol
Thus,
From the reaction below:
3 moles of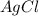 are produced when 1 mole of
are produced when 1 mole of 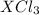 undergoes reaction.
undergoes reaction.
So,
1 mole of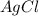 are produced when
are produced when 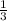 mole of
mole of 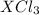 undergoes reaction.
undergoes reaction.
0.01172 mole of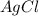 are produced when
are produced when 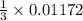 mole of
mole of 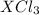 undergoes reaction.
undergoes reaction.
Thus, moles of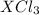 = 0.0039 moles
= 0.0039 moles
Let the atomic mass of X = x g/mol
atomic mass of chlorine = 35.5 g/mol
Thus, Molar mass of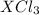 = x + 3(35.5) g/mol = x + 106.5 g/mol
= x + 3(35.5) g/mol = x + 106.5 g/mol
Moles = 0.0039 moles
Mass = 0.521 g
Thus, molar mass = Given mass/ Moles = 0.521 / 0.0039 = 133.5897 g/mol
So,
x + 106.5 = 133.5897
x = 27.0897 g/mol
This Atomic weight corresponds to Aluminum. Hence, X is aluminum.
Ответ: