cristinanina
25.02.2020 •
Chemistry
After doing multiple titrations, your NaOH solution is determined to have a mean concentration value of 0.100 M. Given you are to assume your unknown acid is 75.0% KHP, how many grams of your unknown will you need to use 15.00 mL of your 0.100 M standardized NaOH
Solved
Show answers
More tips
- F Family and Home How to Build a Strong Relationship with Your Child: Tips for Effective Communication...
- H Health and Medicine Boosting Immunity: A Complete Guide on How to Improve Your Body’s Natural Defenses...
- C Computers and Internet The Best Antivirus Programs for your PC...
- S Style and Beauty How to Get Rid of Acne: Scientifically Proven Methods...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
Answers on questions: Chemistry
- C Chemistry Describe the evidence that supports the idea that particles have a property we call charge...
- C Chemistry Check my answers? (image attached)...
- C Chemistry How many types of ions must be present in solution, at minimum, for the phenomenon of selective precipitation to be observed? Your answer should be a whole number without...
- C Chemistry What are the names of the varibles that a scientist uses when performing a scientif investigation experiment)?...
- C Chemistry A0.04336 g sample of gas occupies 10.0-ml at 293.5 k and 1.10 atm. upon further analysis, the compound is found to be 25.305% c and 74.695% cl. what is the molecular formula...
- C Chemistry Lower ionization energy means that it is easier or harder to remove an electron...
- C Chemistry Write an equation for: alpha a decay of radon - 198 beta b decay of uranium - 237 plutonium - 244 undergoes gamma decay !...
- C Chemistry What is the wavelength of the matter wave associated with an electron (me= 9.1 x 10-31 kg) moving with a speed of 2.5 x 107 m/s? a. 2.41*10^-34b.1.51*10^-56c.2.9*10^-11d.1.82...
- P Physics Giúp mình với ạ…mình đang gấp…...
- E English Make this into a parallel sentence… During this event you can also get... Get special items that will set your hands and feet on FIRE! * Juggle Dozens of flame torch with...

Ответ:
The mass of unknown acid needed is 0.230 grams
Explanation:
To calculate the number of moles for given molarity, we use the equation:
Molarity of NaOH solution = 0.100 M
Volume of solution = 15.00 mL = 0.015 L (Conversion factor: 1 L = 1000 mL)
Putting values in above equation, we get:
The chemical reaction for the reaction of KHP and NaOH follows
By Stoichiometry of the reaction:
1 mole of NaOH reacts with 1 mole of KHP.
So, 0.0015 moles of NaOH will react with =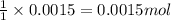 of KHP
of KHP
To calculate the number of moles, we use the equation:Moles of KHP = 0.0015 moles
Molar mass of KHP = 204.22 g/mol
Putting values in above equation, we get:
We are given:
Mass of unknown acid = 75 % of Mass of KHP
So, mass of unknown acid =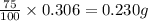
Hence, the mass of unknown acid needed is 0.230 grams
Ответ:
Steel: metallic
Propane: covalent
Calcium chloride: ionic
Water: covalent
Explanation: