halfpint1168
28.06.2019 •
Chemistry
Agalvanic cell has the overall reaction: zn(s) 2eu(no3)3(aq) zn(no3)2(aq) 2eu(no3)2(aq) which is the half reaction occurring at the anode
Solved
Show answers
More tips
- F Food and Cooking How to salt lard?...
- W Work and Career Can Skill Alone Make You a Professional?...
- C Computers and Internet How to Top Up Your Skype Account Without Losing Money?...
- P Philosophy Unidentified Flying Object - What is the Nature of this Phenomenon?...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
Answers on questions: Chemistry
- H History According to Galileo why do some people find his teachings heretical...
- E English How do you write a letter to an invitation to a Celebrity?...
- M Mathematics What is the area of a square computer screen with ow much water can be held by a cylindrical tank with a radius of 12 feet and a height of 30 feet? 21-in. sides?...
- S Social Studies The efforts of marginalized groups in the territory of former european colonies to have more of an influence on archaeological interpretation, research questions, and the management...

Ответ:
The half reaction occurring at anode is
Explanation:
In a chemical cell, substance getting oxidized always act as anode and the one getting reduced always act as cathode.
Oxidation reaction is defined as the reaction in which an atom looses its electrons. Here, oxidation state of the atom increases.
Reduction reaction is defined as the reaction in which an atom gains electrons. Here, the oxidation state of the atom decreases.
For the given chemical cell reaction:
Oxidation half reaction (anode):
Reduction half reaction (cathode):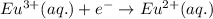 ( × 2 )
( × 2 )
Here, zinc is loosing electrons and is getting oxidized and europium is gaining electrons and loosing electrons.
Hence, the half reaction occurring at anode is
Ответ:
Mate