Agas effuses 1.55 times faster than propane (c3h8)at the
same
temperature and pressure.
what is the mass of the gas?
Solved
Show answers
More tips
- C Computers and Internet Discovering the Differences Between iPad Wi-Fi and iPad 3G...
- F Food and Cooking What s the Best Rice for Cooking Plov?...
- F Family and Home How to Remove Fading from Clothes: Tips and Tricks...
- F Food and Cooking How to Make Polendwitsa at Home?...
- F Family and Home Parents or Environment: Who Has the Most Influence on a Child s Upbringing?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- H History Brainliestttme : ) why was the launch of sputnik in 1957 important? -most in the u.s. had not thought the soviets were capable of producing satellite technology. -...
- H History Which term describes the nationalist movement that sought to create a nation-state for the jewish people...
- E English 1./which of the following best identifies the main claim kim develops in the text? question 1 options: a./asian americans should be encouraged to embrace and celebrate...
- B Business Which of the following would you want to do when developing a market marketing position a. position yourself in an untapped customer market b. market yourself as a...

Ответ:
The mass of the gas is 18.3 g/mol.
Explanation:
To calculate the rate of diffusion of gas, we use Graham's Law.
This law states that the rate of effusion or diffusion of gas is inversely proportional to the square root of the molar mass of the gas. The equation given by this law follows:
Squaring both sides and solving for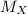
Hence, the molar mas of unknown gas is 18.3 g/mol.
Ответ: