squawk1738
04.12.2019 •
Chemistry
Amixture of nitrogen and hydrogen gases, at a total pressure of 663 mm hg, contains 3.46 grams of nitrogen and 0.156 grams of hydrogen. what is the partial pressure of each gas in the mixture? pn2 = mm hg ph2 = mm hg
Solved
Show answers
More tips
- G Goods and services How to Make Soap at Home: Useful Tips for Beginners...
- C Computers and Internet How to Reinstall Windows: A Detailed Guide for Beginners...
- S Style and Beauty How to Get Rid of a Bruise: Tips and Tricks...
- F Food and Cooking Лечо: вкусное и простое блюдо для любой кухни...
- H Health and Medicine Relieving Swelling in Legs: Causes and Ways to Alleviate the Symptom...
Answers on questions: Chemistry
- C Chemistry 이 Determine the the nuclei indicated in blanks in the right side when Radium (Ra-226) undergoes an alpha decay Ne 226/ 88 Ra -- +...
- M Mathematics any girl can join the meeting no boys allowed I m shreya meeting code:- kxe-rtgw-rqz, in Google meet join fastly it s only for Chatting and don t need to post any irrelevant...
- H History Large numbers of immigrants arrived in the United States between 1820 and 1850. Which groups tended to become farmers on the western frontier?...

Ответ:
The partial pressure of nitrogen gas is 405.76 mmHg and that of hydrogen gas is 257.24 mmHg
Explanation:
To calculate the number of moles, we use the equation:

For nitrogen gas:Given mass of nitrogen gas = 3.46 g
Molar mass of nitrogen gas = 28 g/mol
Putting values in above equation, we get:
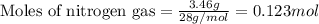
For hydrogen gas:Given mass of hydrogen gas = 0.156 g
Molar mass of hydrogen gas = 2 g/mol
Putting values in above equation, we get:
Mole fraction of a gas is calculated by using the formula:
 ......(1)
......(1)
For nitrogen gas:Putting values in equation 1, we get:

For hydrogen gas:Putting values in equation 1, we get:
The partial pressure of a gas is given by Raoult's law, which is:
where,
 = mole fraction of substance A
= mole fraction of substance A
For nitrogen gas:
For hydrogen gas:Hence, the partial pressure of nitrogen gas is 405.76 mmHg and that of hydrogen gas is 257.24 mmHg
Ответ:
I am expecting that answer is 26600