Amixture of uneven distribution and easy separation is called homogeneous.
a. true
b. false
Solved
Show answers
More tips
- H Health and Medicine Hangover: How to Get Rid of It Quickly?...
- S Style and Beauty How to Choose the Right Fur Coat and Avoid Regrets?...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- C Chemistry ✨free points luv✨ 50 points for each person ;-;...
- C Chemistry The place in the back of your eye that receives images is called the .retina,iris,cornea,pupil...
- C Chemistry What is an example of a physical property?...
- M Mathematics The distance between Mars and Venus is 3.7 X 10^14 miles. A rocket ship only has enough fuel to travel half that distance. How far can the rocket ship travel? PLEASE HELP ME FIRST...
- M Mathematics 30 POINTS FOR ALL OF THESE, BRAINLIEST IF YOU GET THEM ALL CORRECT!!! 1. 30% of 70 equals what percent of 84? A. 35% B. 25% C. 100% D. 40% 2. 75% of 36 equals what percent of 58?...
- M Mathematics Can someone pls help me...

Ответ:
Ответ:
Formula weight of MCl = 58.55 g/mol
M = Sodium
Explanation:
Calculation of the moles of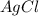 as:-
as:-
Mass = 0.5471 g
Molar mass of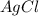 = 143.32 g/mol
= 143.32 g/mol
The formula for the calculation of moles is shown below:
Thus,
1 mole of AgCl is produced from 1 mole of MCl
So, moles of MCl = 0.00381 mol
Mass = 0.2231 grams
Molar mass = Mass/Moles = 0.2231 grams/0.00381 mol= 58.55 g/mol
Molar mass of Cl = 35.5 g/mol
Molar mass of M = Molar mass of MCl - Molar mass of Cl = 58.55 g/mol - 35.5 g/mol = 23.05 g/mol
This mass corresponds to sodium.