Amolecule with the formula ab3 could have one of four different shapes. give the shape and the hybridization of the central a atom for each.
Solved
Show answers
More tips
- C Computers and Internet Can You Insert a Regular SIM Card into an iPad? Let s Find Out...
- F Food and Cooking Лечо: вкусное и простое блюдо для любой кухни...
- H Health and Medicine Relieving Swelling in Legs: Causes and Ways to Alleviate the Symptom...
- S Style and Beauty How to Get Rid of a Bruise: Tips and Tricks...
Answers on questions: Chemistry
- C Chemistry Write the balanced molecular chemical equation for the reaction of aqueous 0.13 m lead (ii) nitrate, with 0.19 m potassium carbonate. you may need to consult appendix...
- C Chemistry The total number of molecules in 34.0 grams of NH3 is equal to A) 2.00 ~ 6.02 x 1023 B) 1.00 6.02 x 1023 C) 1.00 x 22.4 D) 2.00 ~ 22.4...
- C Chemistry calculate the molecular formula of a compound made up of 85.7%c and 14.3% H the molecular mass of compound is 28...
- C Chemistry The particles that make up a liquid move a. more quickly b. more slowly than a gas....
- C Chemistry Ageologist finds a large rock containing a fern fossil. this kind of fern grew on land in hot, moist climates. what does this tell the geologist about the area at the...
- C Chemistry The stage of the cell cycle that follows mitosis is called telophase metaphase interphase cytokinesis...
- C Chemistry Select all that apply to physical and chemical properties. the various components of a mixture do not combine chemically. chemical properties can only be observed during...
- C Chemistry Sedimentary rocks are described as small pieces of rock and animal remains that are weathered, swept downstream and then settle, forming a layer. a. true b. false...
- C Chemistry Which of the following is true of the figures below? a. figure i is a heterogenous mixture, because the particles can be separated and the are not uniformly distributed....
- C Chemistry Extrusive formed igneous rocks cool faster and have larger crystals. a- true b- false...

Ответ:
This molecule is made up of 3 equally spaced sp^2 hybrid orbital arranged at 120 degree angles. The shape of the orbital is planar triangular since there is an atom at the end of each orbital.
Ответ:
The correct answer is Option D.
Explanation:
Lewis dot structures are the structures which represent the valance electrons of an element forming a compound. The electrons are represented as the dots.
In a compound, the bonded electrons are represented as a bond and lone pair of electrons are represented as dots.
In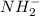 compound,
compound,
Electronic configuration of Nitrogen: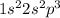
Valence electrons in nitrogen = 5
Electronic configuration of hydrogen: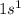
Valence electrons in hydrogen = 1
In the given compound, bond pairs are 2 which means number of electrons bonded are 4.
Unbonded electrons left on nitrogen will be 3 and this compound has an extra electron because of the negative charge present on the compound. Hence, total unbonded electrons present on this compound will be 4.
Therefore, the lewis dot structure will be Option D.