brookeanne723
05.10.2020 •
Chemistry
An object with a mass of 10.0g raises the level of water in a graduated cylinder from 25.0 mL to 30.0 mL. What is the density?
Solved
Show answers
More tips
- C Computers and Internet Protect Your Computer: How to Set a Password on Your Device...
- W Work and Career How to behave at an interview? Tips from an HR specialist...
- F Family and Home How to Keep Your Home Warm: Tips and Tricks...
- D Dating, Love, Relationships Does a Person s Character Depend on the Color of Their Eyes?...
- O Other Childhood Fears: What Many of Us Experienced...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
Answers on questions: Chemistry
- C Chemistry Steel: Propane: Calcium chloride: Water:...
- C Chemistry What can happen when tectonic plates move a few centimeters?...
- C Chemistry The realization that guanine and thymine base pair to cysteine and adenine, respectively, through their keto-like tautomeric forms has ultimately led to which types...
- C Chemistry Review the types of elements that create ionic bonds as well as the types of elements that create covalent bonds. • N2 • CCl4 • SiO2 • AlCl3 • CaCl2 • LiBr...
- C Chemistry The voltage generated by the zinc concentration cell described by, zn(s)|zn2 (aq, 0.100 m)||zn2 (aq, ? m)|zn(s),is 16.0 mv at 25 °c. calculate the concentration...
- C Chemistry What hormone promotes active tubular secretion of potassium ions into, along with sodium absorption from, filtrate in the distal convoluted tubule (dct) and collecting...
- C Chemistry An atom has 8 protons, 8 neutrons, and 8 electrons. another isotope of the same element might have...
- C Chemistry 1. what is an isomer? how many possible isomers of hexane are there? what are the structural differences between these isomers?type your answer here. (score for...
- C Chemistry 3. A vehicle is traveling down the road pictured in the direction of the arrow and is coming to a curve in the road. As the car goes around the curve a passenger...
- P Physics Learning Task 3: Answer the following questions below. Write your answer separate sheet of paper. Among the three pictures A, B, and C, which of the following will...

Ответ:
density=mass/volume
density=10g/5mL
density=2 g/mL
Ответ:
Por definición de molaridad y densidad, la molaridad de la solución es 1.66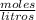 .
.
La concentración molar o molaridad es una medida de la concentración de un soluto en una disolución que indica el número de moles de soluto que están disueltos en un determinado volumen.
La molaridad de una solución se calcula dividiendo los moles del soluto por el volumen de la solución:
La Molaridad se expresa en las unidades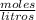 .
.
Por otro lado, la densidad es una magnitud que permite medir la cantidad de masa que hay en determinado volumen de una sustancia.
Entonces, la expresión para el cálculo de la densidad es el cociente entre la masa de un cuerpo y el volumen que ocupa:
En este caso, siendo H₂S el soluto, se colocaron 37 ml del ácido, siendo su densidad 1.36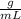 . Reemplazando en la definición de densidad se obtiene:
. Reemplazando en la definición de densidad se obtiene:
masa= 1.36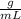 × 37 mL
× 37 mL
masa= 50.32 g
Siendo 34 g/mol la masa molar de H₂S, es decir la cantidad de masa que una sustancia contiene en un mol, la cantidad de moles de H₂S puede ser calculada como:
número de moles de H₂S= 1.48 moles
Siendo 890 mL=0.890 L el volumen de la solución, entonces la molaridad puede ser calculada como:
Molaridad= 1.66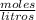
Finalmente, la molaridad de la solución es 1.66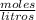 .
.
Aprende más: