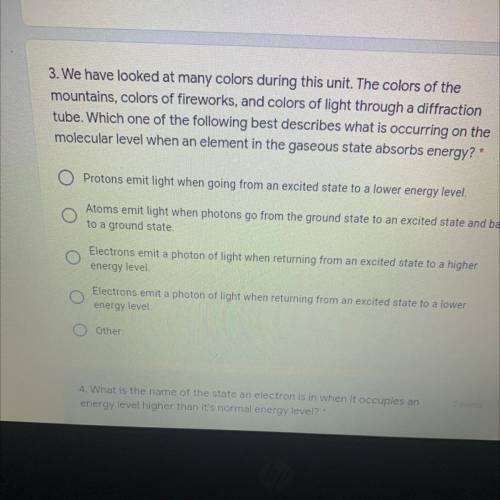
Answer pls i’ll love u
Solved
Show answers
More tips
- C Computers and Internet Discovering the Differences Between iPad Wi-Fi and iPad 3G...
- F Food and Cooking What s the Best Rice for Cooking Plov?...
- F Family and Home How to Remove Fading from Clothes: Tips and Tricks...
- F Food and Cooking How to Make Polendwitsa at Home?...
- F Family and Home Parents or Environment: Who Has the Most Influence on a Child s Upbringing?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- B Business Match each type of insurance with a situation in which the insurance would be useful. 1: when you need to visit a doctor about an illness 2: when your house and possessions are destroyed...
- P Physics Body Mechanics are important for health and fitness, but they also impact sports performance. Paying close attention to how it feels when you are performing a movement can give you...
- B Business In the context of in-house software development options, a firm that enhances a commercial package by adding custom features and configuring it for a particular industry is called...
- A Advanced Placement (AP) during a nuclear reaction, (type of small subatomic particle) strikes the large atomic nucleus of , causing it to break into small pieces and release energy....

Ответ:
are you hot pa.pi?
Explanation:
Ответ:
Explanation: You know that the mass of an individual lead atom is equal to
3.4
⋅
10
−
22
grams.
A substance's density tells you what its mass per unit of volume is. In this case, you know that lead has a density of
11.3 g/cm
3
, which means that each
cm
3
of lead will have a mass of
11.3 g
.
Now, your cube has a volume of
2.00 cm
3
, which means that its mass will be equal to
2.00
cm
3
⋅
density
11.3 g
1
cm
3
=
22.6 g
Now all you have to do is look at the mass of one lead atom and figure out exactly how many lead atoms would be needed to make the total mass of the cube equal to
22.6 g
.
22.6
g
⋅
the mass per atom
1 lead atom
3.4
⋅
10
−
22
g
=
6.647
⋅
10
22
lead atoms
Rounded to two sig figs, the number of sig figs you have for the mass of an individual lead atom, the answer will be
no. of lead atoms
=
6.6
⋅
10
22
atoms
You can double-check this result by using lead's molar mass, which tells you what the exact mass of one mole of lead is.
22.6
g
⋅
1 mole Pb
207.2
g
=
0.1091 moles Pb
Now use Avogadro's number to determine how many atoms of lead you have in that many moles
0.1091
moles
⋅
6.022
⋅
10
23
atoms
1
mole
=
6.57
⋅
10
22
atoms
To two sig figs, you will once again get
no. of lead atoms
=