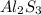ddaaaeeee2960
08.04.2020 •
Chemistry
Apply Concepts
Draw a closed series circuit with two
What would happen if one of the light bulbs bio
les circuit with two light bulbs, a battery, and a switch,
he light bulbs blows out?
Solved
Show answers
More tips
- C Computers and Internet How to Create a Website for Free and Easy?...
- O Other Everything You Need to Know About Kudyabliks...
- C Computers and Internet The Twitter Phenomenon: What it is and How to Use it...
- C Computers and Internet How to Choose a Laptop: Expert Guide and Tips...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...

Ответ:
The empirical formula for the given compound is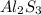
Explanation:
Let us assume that the mass of the compound be 100 grams.
So, the percentage of the elements will be equal to their respective masses.
Mass of aluminium = 35.94 g
Mass of sulfur = 64.06 g
To find the empirical formula for a compound, we follow some steps:
Step 1: Converting the given masses of the elements into moles.To calculate moles, we use the equation:
Moles of aluminium =
Moles of sulfur =
Step 2: Now, dividing each calculated moles by the smallest number of moles, which is 1.33 moles, to calculate the mole ratio of elements.Mole ratio of aluminium =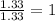
Mole ratio of sulfur =
Step 3: Converting the mole ratio into whole number ratio.Multiplying both the ratios by 2, to make both the mole ratios to be a whole number.
Mole ratio of aluminium =
Mole ratio of sulfur =
Step 4: Writing each mole ratio as the subscripts of the elements to make the empirical formula.Empirical formula for the given compound is