lakhwinder6588
10.12.2019 •
Chemistry
Asample of calcium carbonate (caco3) is known to contain some impurities. it is found that calcium makes up 11% of the entire mass of the sample. all of the calcium comes from the caco3 compound. find the mass percent of caco3 in the sample. set up a ratio and solve.
Solved
Show answers
More tips
- S Science and Technology The Metric System in Our Daily Life: Understanding Its Importance...
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- C Computers and Internet How to Learn to Type Fast?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- S Style and Beauty How to braid friendship bracelets?...
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking Which Calamari Salad is the Most Delicious?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- H Health and Medicine What is Autism? Understanding the Basics of This Neurodevelopmental Disorder...
Answers on questions: Chemistry
- M Mathematics Describe how to solve the inequality 3x+4 13 using a number line....
- M Mathematics The square root of the product of two numbers is the...
- M Mathematics Solve the following inequality x−3 7...
- P Physics Select the correct answer. the image shows street lights powered by solar panels. which sequence shows the energy transformations taking place in these lights? ng place...
- M Mathematics At store A, rulers are sold individually. The cost y of x rulers is represented by the equation y = 0.95x. The costs of rulers at store B are shown in the table....

Ответ:
28.05% is the mass percent of calcium carbonate in the sample.
Explanation:
Suppose in 100 g of sample of impure calcium carbonate
Percentage of calcium in the sample = 11%
Mass of the calcium in the sample = 11% of 100 g =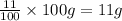
Moles of calcium =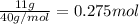
In 1 mole of calcium carbonate there are 1 mol of calcium .Then in 0.275 moles of calcium will be in :
Mass of 0.275 moles of calcium carbonate :
0.275 mol × 102 g/mol =28.05 g
Percentage of calcium carbonate in the sample:
28.05% is the mass percent of calcium carbonate in the sample.
Ответ:
2 acids found on the daily:
-milk (pH level: 6.5) -- consumed through drinking.
-toothpaste (pH level: 6.8) -- used to brush teeth.
2 bases found on the daily:
-hand soap (pH level: 10) -- used to wash hands.
-printing ink (pH level: 9) -- used to print files onto paper, such as printing out school assignments or job resumes.