artemiscrock77041
12.12.2019 •
Chemistry
Asample of sodium bicarbonate is known to contain some impurities. it is found that na makes up 18.00% of the entire mass of the sample. all of the na comes from the nahco3 compound. find the mass percent of nahco3 in the sample
Solved
Show answers
More tips
- H Health and Medicine Flu: How to Recognize It by the First Symptoms?...
- F Food and Cooking How to Sober Up Quickly? Important Facts and Tips...
- H Health and Medicine How to Properly Take a Blood Sugar Test?...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
Answers on questions: Chemistry
- E English To say that language is ambiguous means that we each assign meaning uniquely. language is a random representation. language is guided by rules. language is vague....
- S Spanish 1) how would you say in second period in spanish? en el sexto hora en la sexta hora en el segundo hora en la segunda hora 2) how would you write 1: 30 p.m. using 24 hour time?...
- S Social Studies What are two criticisms of the seniority rule?...
- H History Explain how Andrew Jackson s presidency represented the changing relationship between political candidates and the American people?...

Ответ:
Answer : The mass percent of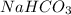 in the sample is, 65.7 %
in the sample is, 65.7 %
Explanation :
As we are given that Na makes up 18.00% of the entire mass of the sample.
First we have to calculate the mass percent of Na in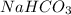 .
.
Mass of Na = 23 g
Mass of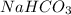 = 84 g
= 84 g
Putting values in above equation, we get:
Now we have to calculate the mass percent of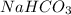 in the sample.
in the sample.
As, 27.4 % amount of Na present in 100 %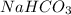
So, 18 % amount of Na present in
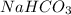
Therefore, the mass percent of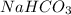 in the sample is, 65.7 %
in the sample is, 65.7 %
Ответ:
A. Having multiple trials.
Explanation: