puppycity78
07.07.2021 •
Chemistry
Assign oxidation state to each atom in each element ion or compound.
a. Ag
b. Ag+
c. CaF2
d. H2S
e.CO3
f. CrO4
g. Cl2
h. Fe
i. CuCl2
j. CH4
Solved
Show answers
More tips
- P Philosophy How did the concept of module arise in computer science?...
- F Food and Cooking How to Cook Julienne? Recipes and Tips...
- H Health and Medicine At What Age Does a Person Stop Growing?...
- F Family and Home How to Choose a Name for Your Baby?...
- F Food and Cooking Discover the Health Benefits of Cedar Nuts...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
- F Food and Cooking How to Make Sushi: A Step-by-Step Guide to Perfectly Rolled Delights...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- F Food and Cooking Аэрогриль: Все, что нужно знать для подготовки здоровой и вкусной пищи...
Answers on questions: Chemistry
- E Engineering How can i solve this? please help...
- M Mathematics Is 313756 divisible by 10?...
- H History In your response, you will be assessed on the following. Respond to the prompt with a historically defensible thesis or claim that establishes a line of reasoning. • Describe...
- M Mathematics A discount movie theater charges $3 for children and $5 for adults. On a particular weekend, 1500 tickets were sold and $5600 was made. How many children and adult tickets were...

Ответ:
a.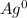
b.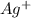
c.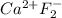
d.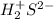
e.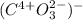
f.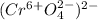
g.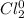
h.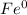
i.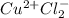
j.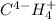
Explanation:
Hello there!
In this case, according to the concept of charge balance, which tell us that the overall charge is zero for any compound, except ions, it turns out possible to proceed as follows:
a.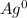
b.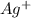
c.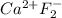
d.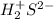
e.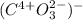
f.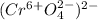
g.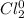
h.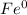
i.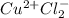
j.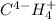
Keep in mind lonely elements have 0 as their oxidation state.
Regards!
Ответ:
The interaction between the two atoms of H in H2 with the lower energy corresponds to a covalent bond between hydrogen's.
When the two atoms of H form a bond, they are overlapping the individual orbitals to form a new one. Every hydrogen has 1 electron which sits in a 1s orbital and then form one molecular orbital. The energy of H2 is lower than individual hydrogens because the electrostatic interaction between them.