Heavenleigh302
10.03.2020 •
Chemistry
Assume that for the pH 9.2 phenolphthalein sample, you used half as much phenolphthalein solution (i.e your sample is 9 ml buffer, 0.5 ml phenophthalein solution and 0.5 ml water). what should happen to the value of Kc you determine?
Solved
Show answers
More tips
- S Style and Beauty How to Properly Tie a Tie: 5 Simple Steps...
- L Leisure and Entertainment How to Make a Crab Trap in Just a Few Minutes...
- H Health and Medicine How Much Does Abortion Cost? Expert Answers and Insights...
- S Sport How to Build Arm Muscles? Effective Exercises and Tips...
- H Health and Medicine When can it be said that a person has a normal pulse?...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
Answers on questions: Chemistry
- C Chemistry 180ft/day is how many cm/hr? must show work w a conversion table pls :)...
- C Chemistry What mass of ethanol will All a 250.0 ml container if its density is 0.789 g/ml?...
- C Chemistry 12. (01.06 LC) Which of the following is a property of a pure substance? (5 points) It consists of different types of compounds. It may have substances in different...
- C Chemistry 28. Which sample has the least number of atoms? A. 1 mol of H2SO4 B. 1 mol of CH3COOH C. 2 mol of H2O2 D. 2 mol of NH3...
- C Chemistry Jane s partner Taylor gives her a ring for valentine s day, which they said is white gold. Jane s friend Sofia thinks it s actually silver. Design an experiment that...
- C Chemistry A. Draw the Lewis structure for ethene (ethylene), H2CCH2, a small hydrocarbon with a C=C double bond. b. Based on this structure, predict the H–C–H bond angle. Explain...
- C Chemistry BRAINLIEST ASAP Draw the electric field lines between a proton and an electron. According to Coulomb s law, how would the electrical force between these particles change...
- C Chemistry What light produces proton bonds to vibrate and heat up?...
- C Chemistry An ice sculpture melts into a puddle of water. what is true of this reaction? it is spontaneous. energy had to be added to the system for the reaction to occur. the...
- C Chemistry Select the word from the list that best fits the definition creating a study group...

Ответ:
Explanation:
It is given that pH is equal to 9.2. So, we will calculate the value of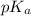 as follows.
as follows.
pH =![pK_{a} + log \frac{[H_{3}O^{+}]}{[\text{solution}]}](/tpl/images/0539/4001/906ab.png)
Putting the given values into the above formula as follows.
pH =![pK_{a} + log \frac{[H_{3}O^{+}]}{[\text{solution}]}](/tpl/images/0539/4001/906ab.png)
= 7.9
=
Since, the value of comes out to be positive. This means that there will occur an increase in the value of
comes out to be positive. This means that there will occur an increase in the value of  or
or 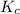 .
.
Ответ:
Imagine a chemist is in the lab and trying to make some chemical reactions happen. In one reaction she reacts chemicals in an exothermic reaction and there is an increase in entropy. A second chemical reaction she is trying to run is endothermic and there is a decrease in entropy. Which of the two reactions is more likely to occur and why?